13.3
Impact Factor
Theranostics 2018; 8(14):3781-3796. doi:10.7150/thno.24821 This issue Cite
Research Paper
Nano-delivery of fraxinellone remodels tumor microenvironment and facilitates therapeutic vaccination in desmoplastic melanoma
1. Division of Pharmacoengineering and Molecular Pharmaceutics and Center for Nanotechnology in Drug Delivery, Eshelman School of Pharmacy, University of North Carolina at Chapel Hill, Chapel Hill, NC 27599, USA.
2. School of Pharmaceutical Sciences, Zhengzhou University, 100 Kexue Avenue, Zhengzhou 450001, China.
3. Collaborative Innovation Center of New Drug Research and Safety Evaluation, Henan Province, China.
4. Key Laboratory of Targeting and Diagnosis for Critical Diseases, Henan Province, China.
5. Department of Environmental Toxicology, The Institute of Environmental and Human Health (TIEHH) and the Center for Biotechnology & Genomics, Texas Tech University, Lubbock, TX 79416, USA.
# These authors contributed equally
Abstract
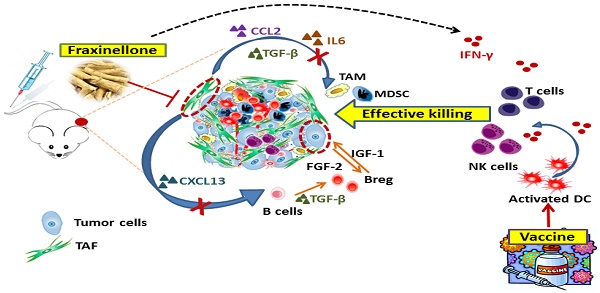
Rationale: Tumor-associated fibroblasts (TAFs) play a critical role in the suppressive immune tumor microenvironment (TME), compromising the efficacy of immunotherapy. To overcome this therapeutic hurdle, we developed a nanoemulsion (NE) formulation to deliver fraxinellone (Frax), an anti-fibrotic medicine, to TAFs, as an approach to reverse immunosuppressive TME of desmoplastic melanoma.
Methods: Frax NE was prepared by an ultrasonic emulsification method. The tumor inhibition effect was evaluated by immunofluorescence staining, masson trichrome staining and western blot analysis. Immune cell populations in tumor and LNs were detected by flow cytometry.
Results: This Frax NE, with a particle size of around 145 nm, can efficiently accumulate in the tumor site after systemic administration and was taken up by TAFs and tumor cells. A significant decrease in TAFs and stroma deposition was observed after intravenous administration of Frax NE, and Frax NE treatment also remolded the tumor immune microenvironment, as was reflected by an increase of natural-killer cells, cytotoxic T cells (CTLs) as well as a decrease of regulatory B cells, and myeloid-derived suppressor cells in the TME. In addition, after treatment by Frax NEs, T helper 1 (Th1) cytokines of interferon gamma (IFN-γ), which effectively elicit anti-tumor immunity, were enhanced. Transforming growth factor-β (TGF-β), chemokine (C-C motif) ligand 2 (CCL2) and interleukin 6 (IL6), which inhibit the development of anti-tumor immunity, were reduced. Although Frax NE demonstrated an inhibitory effect on tumor growth, this mono-therapy could only achieve partial antitumor efficacy, and the tumor growth effect was not maintained long-term after dosing stopped. Therefore, a tumor-specific peptide vaccine was combined with Frax NEs. The combination led to enhanced tumor-specific T-cell infiltration, activated death receptors on the tumor cell surface, and induced increased apoptotic tumor cell death.
Conclusion: Collectively, Frax NE combined with tumor-specific peptide vaccine might be an effective and safe strategy to remodel fibrotic TME, thereby enhancing immune response activation, resulting in a prolonged efficiency for advanced desmoplastic melanoma.
Keywords: fraxinellone, nanoemulsion, desmoplastic melanoma, tumor microenvironment, tumor-associated fibroblasts
 Global reach, higher impact
Global reach, higher impact