13.3
Impact Factor
Theranostics 2019; 9(14):4182-4191. doi:10.7150/thno.34411 This issue Cite
Research Paper
Determination of cerebrospinal fluid leakage by selective deletion of transferrin glycoform using an immunochromatographic assay
1. Department of Chemistry, School of Physics and Chemistry, Gwangju Institute of Science and Technology (GIST), Gwangju 500-712, Republic of Korea
2. Department of Chemical and Biological Engineering, Rensselaer Polytechnic Institute, Troy, NY, USA
3. Center for Biotechnology and Interdisciplinary Studies, Rensselaer Polytechnic Institute, Troy, NY, USA
4. Department of Chemistry and Chemical Biology, Rensselaer Polytechnic Institute, Troy, NY, USA
5. Division of Neurosurgery, Department of Neuroscience NYU-Winthrop Hospital, 259 1st St Mineola, NY 11501 and MD in Neurological Surgery P.C. Rockville Center, New York
Abstract
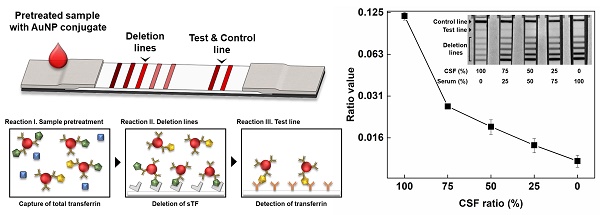
Cerebrospinal fluid (CSF) leakage can lead to brain and spine pathologies and there is an urgent need for a rapid diagnostic method for determining CSF leakage. Beta-2 transferrin (β2TF), asialotransferrin, is a specific CSF glycoprotein biomarker used to determine CSF leakage when distinguished from serum sialotransferrin (sTF).
Methods: We detected β2TF using an immunochromatographic assay (ICA), which can be potentially developed as a point-of-care (POC) testing platform. Sialic acid-specific lectin selectively captures sTF in multiple deletion lines within an ICA test strip, enabling the detection of β2TF. A sample pre-treatment process efficiently captures excess sTF increasing sensitivity for CSF leakage detection.
Results: An optimal cut-off value for determining the presence of CSF in test samples was obtained from receiver operating characteristic (ROC) analysis of the ratio of the test signal intensity and the deletion lines. On 47 clinical samples, ICA test strips discriminated CSF positive from negative samples with statistically significant (positive versus negative t-test; P =0.00027). Additional artificial positive samples, prepared by mixing CSF positive and negative clinical samples, were used as a further challenge. These positive samples were clearly discriminated from the negative samples (mixture versus negative t-test; P =0.00103) and CSF leakage was determined with 97.1% specificity and 96.2% sensitivity.
Conclusions: ICA represents a promising approach for POC diagnosis of CSF leakage. While requiring 70 min assay time inconvenient for POC testing, our method was significantly shorter than conventional electrophoresis-based detection methods for β2TF.
Keywords: cerebrospinal fluid leakage, transferrin, sialic acid, immunochromatographic assay, point of care testing
 Global reach, higher impact
Global reach, higher impact