Impact Factor
Theranostics 2019; 9(4):1001-1014. doi:10.7150/thno.30056 This issue Cite
Research Paper
Tumor-secreted dickkopf2 accelerates aerobic glycolysis and promotes angiogenesis in colorectal cancer
1. Department of Pathology, Nanfang Hospital, Southern Medical University, Guangzhou, China
2. Department of Pathology, School of Basic Medical Sciences, Southern Medical University, Guangzhou, China
3. Gastrointestinal Surgical Center, the First Affiliated Hospital of Sun Yat-sen University, Guangzhou, China
4. Department of Medical Oncology, Affiliated Tumor Hospital of Guangzhou Medical University, Guangzhou, China
5. Department of General Surgery, Zhujiang Hospital, Southern Medical University, Guangzhou, China
6. Department of General Surgery, Guangdong General Hospital, Guangdong Academy of Medical Science, Guangzhou, China.
7. Second Department of Hepatobiliary Surgery, Zhujiang Hospital, Southern Medical University, Guangzhou, China
* These authors are contributed equally to this work
Abstract
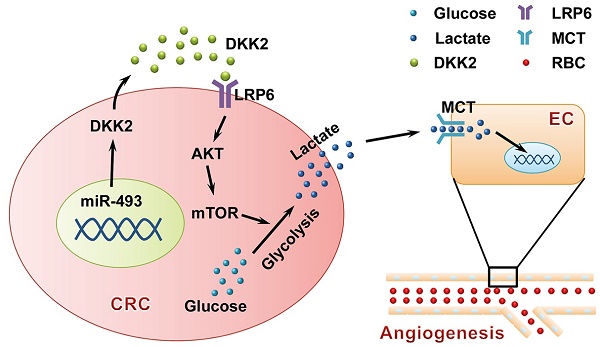
Angiogenesis is a fundamental process that involves in tumor progression and metastasis. Vascular endothelial growth factor (VEGF) family and their receptors are identified as the most prominent regulators of angiogenesis. However, the clinical efficacy of anti-VEGF/VEGFR therapy is not ideal, prompting the needs to further understand mechanisms behind tumor angiogenesis. Here, we found that Dickkopf associated protein 2 (DKK2), a secretory protein highly expressed in metastatic colorectal cancer tissues, could stimulate angiogenesis via a classic VEGF/VEGFR independent pathway.
Methods: DKK2 was screened out from microarray data analyzing gene expression profiles of eight pairs of non-metastatic and metastatic human colorectal cancer (CRC) tissues. Immunofluorescence histochemical staining (IHC) was used to detect the expression of DKK2 and angiogenesis in CRC tissues. Chicken chorioallantoic membrane (CAM) assay and Human umbilical vein endothelial cells (HUVEC) tubule formation assay was used for in vitro and in vivo angiogenesis study, respectively. Lactate and glucose concentration in the culture medium was measured by enzyme-linked immunosorbent assay (ELISA). Luciferase reporter assay was used to verify the interaction between miR-493-5p and the 3'UTR of DKK2.
Results: DKK2 could stimulate angiogenesis via accelerating the aerobic glycolysis of CRC cells, through which lactate is produced from glucose and accumulated in tumor microenvironment. Lactate functions as the final executor of DDK2 to stimulate tube formation of endothelial cells, and blockage of lactate secretion by lactate transporter (MCT) inhibitors dramatically neutralize the progression and metastasis of CRC both in vitro and in vivo. DKK2 could cooperate with lipoprotein receptor-related protein 6, which is required for glucose uptake, and activated the downstream mTOR signal pathway to accelerate lactate secretion. In addition, the expression of DKK2 is switched on via the demethylation of miR-493-5p, which allows the dissociated of miR-493-5p from the 3′-UTRs of DKK2 and initiates its stimulatory role on CRC progression in an autocrine or paracrine manner.
Conclusion: DKK2 promotes tumor metastasis and angiogenesis through a novel VEGF-independent, but energy metabolism related pathway. DKK2 might be a potential anti-angiogenic target in clinical treatment for the advanced CRC patients.
Keywords: Colorectal cancer, Vascular endothelial growth factor, Dickkopf 2, Aerobic Glycolysis, Angiogenesis
 Global reach, higher impact
Global reach, higher impact