13.3
Impact Factor
Theranostics 2018; 8(17):4765-4780. doi:10.7150/thno.25162 This issue Cite
Research Paper
Simultaneous characterization of tumor cellularity and the Warburg effect with PET, MRI and hyperpolarized 13C-MRSI
1. Department of Nuclear Medicine, Klinikum rechts der Isar, Technical University of Munich, 81675 München, Germany
2. Department of Chemistry, Technical University of Munich, 85748 Garching, Germany
3. Department of Radiology, Medical Physics, University Medical Center Freiburg, Faculty of Medicine, University of Freiburg, 79106 Freiburg, Germany
4. German Consortium for Cancer Research (DKTK), 69120 Heidelberg, Germany
5. German Cancer Research Center (DKFZ), 69120 Heidelberg, Germany
6. Department of Clinical Physiology, Nuclear Medicine & PET and Cluster for Molecular Imaging Rigshospitalet and University of Copenhagen, 2100 Copenhagen, Denmark
7. Department of Radiology, Klinikum rechts der Isar, Technical University of Munich, 81675 München, Germany
8. Munich School of Bioengineering, Technical University of Munich, 85748 Garching, Germany
9. Department of Pathology, Klinikum rechts der Isar, Technical University Munich, 81675 München, Germany
10. Comparative Experimental Pathology, Klinikum rechts der Isar, Technical University Munich, 81675 München, Germany
11. Section for Biomedical Imaging, Molecular Imaging North Competence Center (MOINCC), Department for Radiology and Neuroradiology, University Medical Center Kiel, University Kiel
*equal author contribution
Abstract
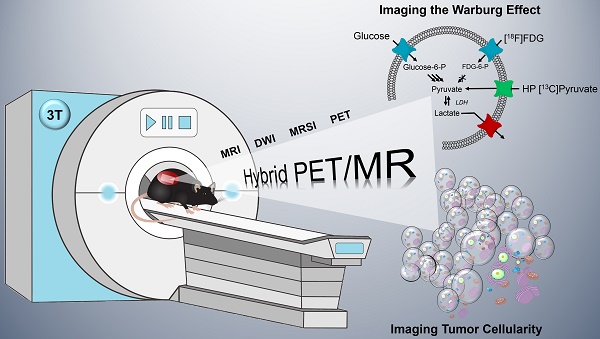
Modern oncology aims at patient-specific therapy approaches, which triggered the development of biomedical imaging techniques to synergistically address tumor biology at the cellular and molecular level. PET/MR is a new hybrid modality that allows acquisition of high-resolution anatomic images and quantification of functional and metabolic information at the same time. Key steps of the Warburg effect-one of the hallmarks of tumors-can be measured non-invasively with this emerging technique. The aim of this study was to quantify and compare simultaneously imaged augmented glucose uptake and LDH activity in a subcutaneous breast cancer model in rats (MAT-B-III) and to study the effect of varying tumor cellularity on image-derived metabolic information.
Methods: For this purpose, we established and validated a multimodal imaging workflow for a clinical PET/MR system including proton magnetic resonance (MR) imaging to acquire accurate morphologic information and diffusion-weighted imaging (DWI) to address tumor cellularity. Metabolic data were measured with dynamic [18F]FDG-PET and hyperpolarized (HP) 13C-pyruvate MR spectroscopic imaging (MRSI). We applied our workflow in a longitudinal study and analyzed the effect of growth dependent variations of cellular density on glycolytic parameters.
Results: Tumors of similar cellularity with similar apparent diffusion coefficients (ADC) showed a significant positive correlation of FDG uptake and pyruvate-to-lactate exchange. Longitudinal DWI data indicated a decreasing tumor cellularity with tumor growth, while ADCs exhibited a significant inverse correlation with PET standard uptake values (SUV). Similar but not significant trends were observed with HP-13C-MRSI, but we found that partial volume effects and point spread function artifacts are major confounders for the quantification of 13C-data when the spatial resolution is limited and major blood vessels are close to the tumor. Nevertheless, analysis of longitudinal data with varying tumor cellularity further detected a positive correlation between quantitative PET and 13C-data.
Conclusions: Our workflow allows the quantification of simultaneously acquired PET, MRSI and DWI data in rodents on a clinical PET/MR scanner. The correlations and findings suggest that a major portion of consumed glucose is metabolized by aerobic glycolysis in the investigated tumor model. Furthermore, we conclude that variations in cell density affect PET and 13C-data in a similar manner and correlations of longitudinal metabolic data appear to reflect both biochemical processes and tumor cellularity.
Keywords: PET/MR, [18F]FDG-PET, hyperpolarized 13C-MRSI, DNP, diffusion-weighted imaging, multimodal imaging, NMR, spectroscopy
 Global reach, higher impact
Global reach, higher impact