13.3
Impact Factor
Theranostics 2018; 8(16):4295-4304. doi:10.7150/thno.26370 This issue Cite
Research Paper
Comparative biodistribution analysis across four different 89Zr-monoclonal antibody tracers—The first step towards an imaging warehouse
1. Department of Medical Oncology, University of Groningen, University Medical Center Groningen, the Netherlands.
2. Department of Clinical Pharmacy and Pharmacology, University of Groningen, University Medical Center Groningen, the Netherlands.
3. Department of Nuclear Medicine and Molecular Imaging, University of Groningen, University Medical Center Groningen, the Netherlands.
4. Department of Medical Oncology, Cancer Center Amsterdam, VU University Medical Center, Amsterdam, the Netherlands.
Abstract
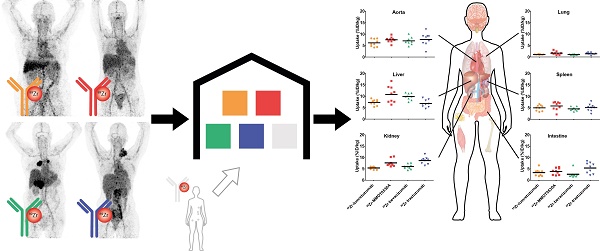
Rationale: Knowledge on monoclonal antibody biodistribution in healthy tissues in humans can support clinical drug development. Molecular imaging with positron emission tomography (PET) can yield information in this setting. However, recent imaging studies have analyzed the behavior of single antibodies only, neglecting comparison across different antibodies.
Methods: We compared the distribution of four 89Zr-labeled antibodies in healthy tissue in a retrospective analysis based on the recently published harmonization protocol for 89Zr-tracers and our delineation protocol.
Results: The biodistribution patterns of 89Zr-lumretuzumab, 89Zr-MMOT0530A, 89Zr-bevacizumab and 89Zr-trastuzumab on day 4 after tracer injection were largely similar. The highest tracer concentration was seen in healthy liver, spleen, kidney and intestines. About one-third of the injected tracer dose was found in the circulation, up to 15% in the liver and only 4% in the spleen and kidney. Lower tracer concentration was seen in bone marrow, lung, compact bone, muscle, fat and the brain. Despite low tracer accumulation per gram of tissue, large-volume tissues, especially fat, can influence overall distribution: On average, 5-7% of the injected tracer dose accumulated in fat, with a peak of 19% in a patient with morbid obesity.
Conclusion: The similar biodistribution of the four antibodies is probably based on their similar molecular structure, binding characteristics and similar metabolic pathways. These data provide a basis for a prospectively growing, online accessible warehouse of molecular imaging data, which enables researchers to increase and exchange knowledge on whole body drug distribution and potentially supports drug development decisions.
Keywords: PET, Zirconium-89, monoclonal antibody, biodistribution
 Global reach, higher impact
Global reach, higher impact