13.3
Impact Factor
Theranostics 2018; 8(14):3751-3765. doi:10.7150/thno.22493 This issue Cite
Research Paper
Identification and Functional Characterization of Long Non-coding RNA MIR22HG as a Tumor Suppressor for Hepatocellular Carcinoma
1. Department of Radiation Oncology, Nanfang Hospital, Southern Medical University, Guangzhou 510515, China
2. State Key Laboratory of Organ Failure Research, Nanfang Hospital, Southern Medical University, Guangzhou 510515, China
3. Guangdong Provincial Key Laboratory of Viral Hepatitis Research, Nanfang Hospital, Southern Medical University, Guangzhou 510515, China
4. Department of Infectious Diseases, Nanfang Hospital, Southern Medical University, Guangzhou 510515, China
5. Department of General Surgery, Nanfang Hospital, Southern Medical University, Guangzhou 510515, China
† These authors contributed equally to this work.
Abstract
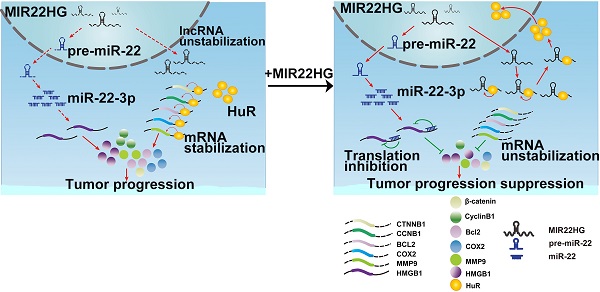
Long non-coding RNAs (lncRNAs) have recently been identified as critical regulators in tumor initiation and development. However, the function of lncRNAs in human hepatocellular carcinoma (HCC) remains largely unknown. Our study was designed to explore the biological function and clinical implication of lncRNA MIR22HG in HCC.
Methods: We evaluated MIR22HG expression in 52-patient, 145-patient, TCGA, and GSE14520 HCC cohorts. The effects of MIR22HG on HCC were analyzed in terms of proliferation, invasion, and metastasis, both in vitro and in vivo. The mechanism of MIR22HG action was explored through bioinformatics, luciferase reporter, and RNA immunoprecipitation analyses.
Results: MIR22HG expression was significantly down-regulated in 4 independent HCC cohorts compared to that in controls. Its low expression was associated with tumor progression and poor prognosis of patients with HCC. Forced expression of MIR22HG in HCC cells significantly suppressed proliferation, invasion, and metastasis in vitro and in vivo. Mechanistically, MIR22HG derived miR-22-3p to target high mobility group box 1 (HMGB1), thereby inactivating HMGB1 downstream pathways. Additionally, MIR22HG directly interacted with HuR and regulated its subcellular localization. MIR22HG competitively bound to human antigen R (HuR), resulting in weakened expression of HuR-stabilized oncogenes, such as β-catenin. Furthermore, miR-22-3p suppression, HuR or HMGB1 overexpression rescued the inhibitory effects caused by MIR22HG overexpression.
Conclusion: Our findings revealed that MIR22HG plays a key role in tumor progression by suppressing the proliferation, invasion, and metastasis of tumor cells, suggesting its potential role as a tumor suppressor and prognostic biomarker in HCC.
Keywords: liver cancer, miR-22 host gene, HuR, tumor suppressor, prognostic factor
 Global reach, higher impact
Global reach, higher impact