13.3
Impact Factor
Theranostics 2018; 8(6):1723-1739. doi:10.7150/thno.22056 This issue Cite
Research Paper
The enhanced antitumor-specific immune response with mannose- and CpG-ODN-coated liposomes delivering TRP2 peptide
1. International Nanobody Research Center of Guangxi, Guangxi Medical University, Nanning, Guangxi, 530021, China
2. The Department of Immunology, Guangxi Medical University, Nanning, Guangxi, 530021, China
3. College of Stomatology, Guangxi Medical University (GXMUCS), Nanning, Guangxi, 530021, China
* These authors contributed equally to this work.
Abstract
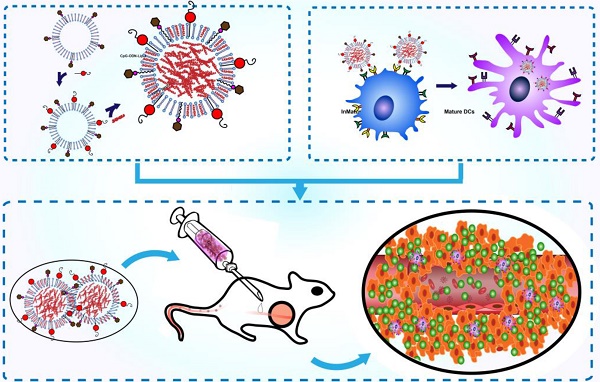
Purpose: Dendritic cell (DC)-based cancer vaccines is a newly emerging and potent form of immune therapy. As for any new technology, there are still considerable challenges that need to be addressed. Here, we investigate the antitumor potential of a novel liposomal vaccine, M/CpG-ODN-TRP2-Lipo.
Methods: We developed a vaccination strategy by assembling the DC-targeting mannose and immune adjuvant CpG-ODN on the surface of liposomes, which were loaded with melanoma-specific TRP2180-188 peptide as liposomal vaccine. M/CpG-ODN-TRP2-Lipo treatment was used to intendedly induce activation of DCs and antitumor- specific immune response in vivo.
Results: Our results demonstrated in vitro that the prepared liposomal particles were efficiently taken up by DCs. This uptake led to an enhanced activation of DCs, as measured by the upregulation of MHC II, CD80, and CD86. Furthermore, M/CpG-ODN-TRP2-Lipo effectively inhibited the growth of implanted B16 melanoma and prolonged the survival of mice. This therapy significantly reduced the number of myeloid-derived suppressor cells (MDSCs) and regulatory T cells, while simultaneously increasing the number of activated T cells, tumor antigen-specific CD8+ cytotoxic T cells, and interferon-γ-producing cells. At the same time, it was found to suppress tumor angiogenesis and tumor cell proliferation, as well as up-regulate their apoptosis. Interestingly, MyD88-knockout mice had significantly shorter median survival times compared to wild-type mice following the administration of M/CpG-ODN-TRP2-Lipo.
Conclusions: The results suggested that the antitumor activities of the vaccine partially rely on the Myd88 signaling pathway. Interestingly, compared to whole tumor cell lysate-based vaccine, M/CpG-ODN-TRP2-Lipo, tumor specific antigen peptide-based vaccine, improved survival of tumor-bearing mice as well as enhanced their antitumor responses. All in all, we describe a novel vaccine formulation, M/CpG-ODN-TRP2-Lipo, with the aim of improving antitumor responses by alleviating the immunosuppressive environment in tumors.
Keywords: immunotherapy, liposome, tumor, dendritic cells-based cancer vaccine, tumor antigen-specific CD8+ T-cells
 Global reach, higher impact
Global reach, higher impact