Impact Factor
Theranostics 2018; 8(6):1690-1705. doi:10.7150/thno.21575 This issue Cite
Research Paper
E. coli Nissle 1917-Derived Minicells for Targeted Delivery of Chemotherapeutic Drug to Hypoxic Regions for Cancer Therapy
1. Hunan Provincial Key Laboratory of Microbial Molecular Biology, State Key Laboratory of Developmental Biology of Freshwater Fish, College of Life Science, Hunan Normal University, Changsha 410081, P.R. China;
2. Department of Medical Imaging, Jinling Hospital, School of Medicine, Nanjing University, Nanjing 210002 Jiangsu, P.R. China.
* These authors contributed equally to this work.
Abstract
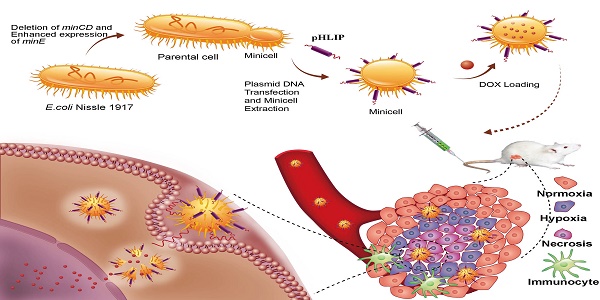
Purpose: Systemic administration of free chemotherapeutic drugs leads to severe toxic effects, and physiological characteristics of solid tumors restrain the drugs from reaching the hypoxic regions. E. coli Nissle 1917 (EcN) has been known to penetrate the barrier and proliferate in the interface between the viable and necrotic regions of tumors. This study aimed to fabricate a nanoscale minicell via genetic engineering of EcN for targeted delivery of chemotherapeutic drugs to the hypoxic regions of tumors for cancer therapy.
Methods: A large number of minicells were produced by knocking out the minCD gene and enhancing the minE expression in EcN. Then, a pH (low) insertion peptide (pHLIP) was displayed on the membrane surface through protein display technology to endow the cells with the ability to target the acidic microenvironments of tumors. The acidic-microenvironment targeting ability and therapeutic effect of the engineered minicells with chemotherapeutic drugs was thoroughly evaluated by using breast cancer cells and an orthotopic model of breast tumor.
Results: The EcN-derived minicells displaying pHLIP could be directly extracted from the fermentation broth and used for delivering chemotherapeutic drugs without any further modification. Targeting of doxorubicin (DOX)-loaded minicells to cancer cells via pHLIP resulted in rapid internalization and drug release in acidic media. Importantly, the pHLIP-mosaic minicells successfully invaded the necrotic and hypoxic regions of orthotopic breast cancers where free chemotherapeutic drugs could never get to because of vascular insufficiency and high interstitial fluid pressure. This invasion resulted in significant regression of an orthotopic breast tumor in a mouse model, while no seriously pathogenic effects were observed during the animal experiments.
Conclusions: This study provides a novel strategy for the fabrication of tumor-targeting carriers via genetic engineering based on biomaterials with the ability to penetrate hypoxic regions of tumors, high biocompatibility and low toxicity.
Keywords: E. coli Nissle 1917, minicell, low pH insert protein, hypoxic region, cancer therapy
 Global reach, higher impact
Global reach, higher impact