13.3
Impact Factor
Theranostics 2018; 8(4):1180-1194. doi:10.7150/thno.20846 This issue Cite
Research Paper
LncRNA PFL contributes to cardiac fibrosis by acting as a competing endogenous RNA of let-7d
1. Department of Pharmacology (State-Province Key Laboratories of Biomedicine-Pharmaceutics of China, Key Laboratory of Cardiovascular Research, Ministry of Education), College of Pharmacy, Harbin Medical University, Harbin, Heilongjiang 150081, P. R. China
2. Northern Translational Medicine Research and Cooperation Center, Heilongjiang Academy of Medical Sciences, Harbin Medical University, Harbin, Heilongjiang 150081, P. R. China.
#Authors with equal contribution to this study
Abstract
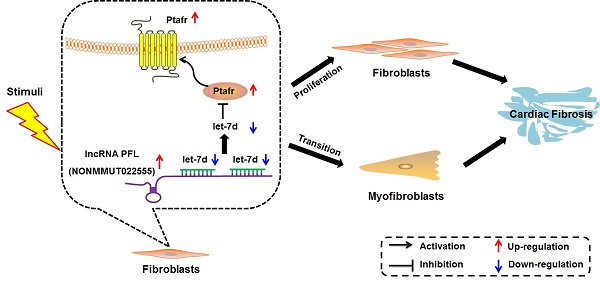
Rationale: Cardiac fibrosis is associated with various cardiovascular diseases and can eventually lead to heart failure. Dysregulation of long non-coding RNAs (lncRNAs) has recently been recognized as one of the key mechanisms involved in cardiac diseases. However, the potential roles and underlying mechanisms of lncRNAs in cardiac fibrosis have not been explicitly delineated.
Methods and Results: Using a combination of in vitro and in vivo studies, we identified a lncRNA NONMMUT022555, which is designated as a pro-fibrotic lncRNA (PFL), and revealed that PFL is up-regulated in the hearts of mice in response to myocardial infarction (MI) as well as in the fibrotic cardiac fibroblasts (CFs). We found that knockdown of PFL by adenoviruses carrying shRNA attenuated cardiac interstitial fibrosis and improved ejection fraction (EF) and fractional shortening (FS) in MI mice. Further study showed that forced expression of PFL promoted proliferation, fibroblast-myofibroblast transition and fibrogenesis in mice CFs by regulating let-7d, whereas silencing PFL mitigated TGF-β1-induced myofibroblast generation and fibrogenesis. More importantly, PFL acted as a competitive endogenous RNA (ceRNA) of let-7d, as forced expression of PFL reduced the expression and activity of let-7d. Moreover, let-7d levels were decreased in the MI mice and in fibrotic CFs. Inhibition of let-7d resulted in fibrogenesis in CFs, whereas forced expression of let-7d abated fibrogenesis through targeting platelet-activating factor receptor (Ptafr). Furthermore, overexpression of let-7d by adenoviruses carrying let-7d precursor impeded cardiac fibrosis and improved cardiac function in MI mice.
Conclusion: Taken together, our study elucidated the role and mechanism of PFL in cardiac fibrosis, indicating the potential role of PFL inhibition as a novel therapy for cardiac fibrosis.
Keywords: cardiac fibrosis, lncRNA PFL, let-7d, Ptafr
 Global reach, higher impact
Global reach, higher impact