13.3
Impact Factor
Theranostics 2017; 7(18):4517-4536. doi:10.7150/thno.19403 This issue Cite
Research Paper
Locoregional Confinement and Major Clinical Benefit of 188Re-Loaded CXCR4-Targeted Nanocarriers in an Orthotopic Human to Mouse Model of Glioblastoma
1. CRCINA, INSERM, Université de Nantes, Université d'Angers, 49933 Angers, France;
2. PACeM, “Plateforme d'Analyses Cellulaires et Moléculaires”, Université d'Angers, 49933 Angers, France;
3. PRIMEX, “Plateforme de Radiobiologie et d'Imagerie EXperimentale”, Université d'Angers, 49933 Angers, France;
4. CRCINA, INSERM, Université d'Angers, Université de Nantes, 44035 Nantes, France;
5. Université Tours, EA6295, “Nanomédicaments et Nanosondes”, 37020 Tours, France;
6. Centre Regional de Lutte Contre le Cancer Eugène Marquis, Department of Nuclear Medicine, 35042 Rennes, France;
7. INSERM U1205, bâtiment modulaire 40-23, CEA 17 rue des Martyrs, 38054 Grenoble cedex, France.
Abstract
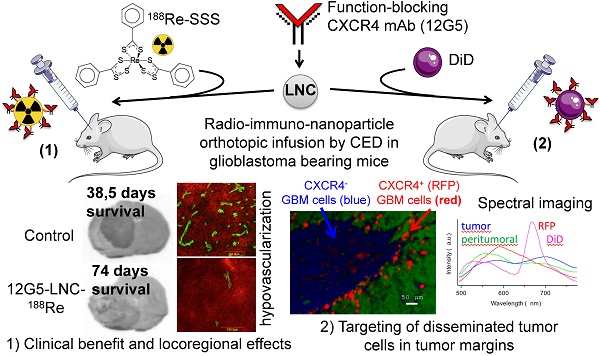
Purpose: Gold standard beam radiation for glioblastoma (GBM) treatment is challenged by resistance phenomena occurring in cellular populations well prepared to survive or to repair damage caused by radiation. Among signals that have been linked with radio-resistance, the SDF1/CXCR4 axis, associated with cancer stem-like cell, may be an opportune target. To avoid the problem of systemic toxicity and blood-brain barrier crossing, the relevance and efficacy of an original system of local brain internal radiation therapy combining a radiopharmaceutical with an immuno-nanoparticle was investigated.
Experiment design: The nanocarrier combined lipophilic thiobenzoate complexes of rhenium-188 loaded in the core of a lipid nanocapsule (LNC188Re) with a function-blocking antibody, 12G5 directed at the CXCR4, on its surface. The efficiency of 12G5-LNC188Re was investigated in an orthotopic and xenogenic GBM model of CXCR4-positive U87MG cells implanted in the striatum of Scid mice.
Results: We demonstrated that 12G5-LNC188Re single infusion treatment by convection-enhanced delivery resulted in a major clinical improvement in median survival that was accompanied by locoregional effects on tumor development including hypovascularization and stimulation of the recruitment of bone marrow derived CD11b- or CD68-positive cells as confirmed by immunohistochemistry analysis. Interestingly, thorough analysis by spectral imaging in a chimeric U87MG GBM model containing CXCR4-positive/red fluorescent protein (RFP)-positive- and CXCR4-negative/RFP-negative-GBM cells revealed greater confinement of DiD-labeled 12G5-LNCs than control IgG2a-LNCs in RFP compartments.
Main conclusion: These findings on locoregional impact and targeting of disseminated cancer cells in tumor margins suggest that intracerebral active targeting of nanocarriers loaded with radiopharmaceuticals may have considerable benefits in clinical applications.
Keywords: CXCR4, glioblastoma, radiation therapy, immuno-targeting, spectral imaging, macrophage, nanoparticle.
 Global reach, higher impact
Global reach, higher impact