13.3
Impact Factor
Theranostics 2017; 7(18):4383-4398. doi:10.7150/thno.20745 This issue Cite
Research Paper
Rational design of Polymeric Hybrid Micelles to Overcome Lymphatic and Intracellular Delivery Barriers in Cancer Immunotherapy
1. Key Laboratory of Drug Targeting and Drug Delivery Systems, Ministry of Education, West China School of Pharmacy, Sichuan University, Chengdu 610041, China;
2. College of Pharmacy and Biological Engineering, Chengdu University, Chengdu 610106, China.
* Contributed equally to this work.
Abstract
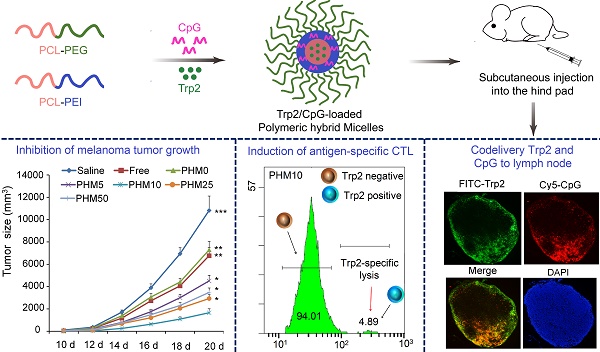
Poor distribution of antigen/adjuvant to target sites and inadequate induction of T cell responses remain major challenges in cancer immunotherapy because of the lack of appropriate delivery systems. Nanocarrier-based antigen delivery systems have emerged as an innovative strategy to improve vaccine efficacy. Here we present polymeric hybrid micelles (PHMs) as a simple and potent antigen/adjuvant co-delivery system with highly tunable properties. PHMs consist of two amphiphilic diblock copolymers, polycaprolactone-polyethylenimine (PCL-PEI) and polycaprolactone-polyethyleneglycol (PCL-PEG). PHMs with different proportions of cationic PCL-PEI were prepared and loaded with tyrosinase-related protein 2 (Trp2) peptide and adjuvant CpG oligodeoxynucleotide to generate the Trp2/PHM/CpG co-delivery system. Lymphatic and intracellular antigen delivery as a function of PCL-PEI proportion was evaluated in vitro and in vivo. PHMs containing 10% (w/w) PCL-PEI (Trp2/PHM10/CpG) showed the optimal balance of good distribution to lymph nodes, strong immunization effect after subcutaneous administration, and low toxicity to dendritic cells. In a mouse model of B16F10 melanoma, Trp2/PHM10/CpG showed significantly higher antigen-specific cytotoxic T lymphocyte activity and greater anticancer efficacy than Trp2/PHM0/CpG without PCL-PEI or a mixture of free Trp2 and CpG. These results provide new insights into how cationic segments affect the efficiency of antigen delivery by cationic nanocarriers. They also suggest that PHMs can serve as a structurally simple and highly tunable platform for co-delivery of antigen and adjuvant in cancer immunotherapy.
Keywords: cancer immunotherapy, co-delivery system, amphiphilic diblock copolymers, tyrosinase-related protein 2 peptide, subcutaneous administration.
 Global reach, higher impact
Global reach, higher impact