13.3
Impact Factor
Theranostics 2017; 7(5):1177-1191. doi:10.7150/thno.17895 This issue Cite
Research Paper
Tumor Hypoxia Regulates Forkhead Box C1 to Promote Lung Cancer Progression
1. Institute of Molecular Biology College of Life Science, National Chung Hsing University, Taiwan;
2. Graduate Institute of Biomedical sciences, China Medical University, Taiwan;
3. Department of Neurology, Center for Neuropsychiatry, and Graduate Institute of Immunology, China Medical University and Hospital, Taichung, Taiwan;
4. National PET/Cyclotron Center and Department of Nuclear Medicine, Taipei Veterans General Hospital, Taipei, Taiwan;
5. Graduate Institute of Biomedical and Pharmaceutical science, Fu Jen Catholic University, New Taipei, Taiwan;
6. Department of Physiology and Pharmacology, Chang Gung University, Tao-Yuan, Taiwan;
7. Department of Neurosurgery, Taichung Veterans General Hospital, Taichung, Taiwan;
8. Department of Medical Research, China Medical University Hospital, Taichung, Taiwan;
9. Department of Biomedical Informatics, Asia University, Taichung, Taiwan.
Abstract
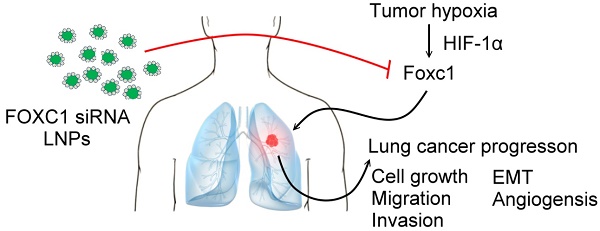
Forkhead box C1 (FOXC1) is a member of the forkhead family of transcription factors that are characterized by a DNA-binding forkhead domain. Increasing evidence indicates that FOXC1 is involved in tumor progression. However, the role of tumor hypoxia in FOXC1 regulation and its impact on lung cancer progression are unclear. Here, we report that FOXC1 was upregulated in hypoxic areas of lung cancer tissues from rodents or humans. Hypoxic stresses significantly induced FOXC1 expression. Moreover, hypoxia activated FOXC1 transcription via direct binding of hypoxia-inducible factor-1α (HIF-1α) to the hypoxia-responsive element (HRE) in the FOXC1 promoter. FOXC1 gain-of-function in lung cancer cells promoted cell proliferation, migration, invasion, angiogenesis, and epithelial-mesenchymal transition in vitro. However, a knockdown of FOXC1 in lung cancer cells inhibited these effects. Notably, knockdown of tumor hypoxia-induced FOXC1 expression via HIF-1-mediated FOXC1 shRNAs in lung cancer xenograft models suppressed tumor growth and angiogenesis. Finally, systemic delivery of FOXC1 siRNA encapsulated in lipid nanoparticles inhibited tumor growth and increased survival time in lung cancer-bearing mice. Taken together, these data indicate that FOXC1 is a novel hypoxia-induced transcription factor and plays a critical role in tumor microenvironment-promoted lung cancer progression. Systemic FOXC1 blockade therapy may be an effective therapeutic strategy for lung cancer.
Keywords: lung cancer, tumor hypoxia, fork head box C1, hypoxia-inducible factor-1α.
 Global reach, higher impact
Global reach, higher impact