13.3
Impact Factor
Theranostics 2017; 7(1):164-179. doi:10.7150/thno.17099 This issue Cite
Research Paper
Smart Cu(II)-aptamer complexes based gold nanoplatform for tumor micro-environment triggered programmable intracellular prodrug release, photodynamic treatment and aggregation induced photothermal therapy of hepatocellular carcinoma
1. The United Innovation of Mengchao Hepatobiliary Technology Key Laboratory of Fujian Province, Mengchao Hepatobiliary Hospital of Fujian Medical University, Fuzhou 350025, P. R. China;
2. The Liver Center of Fujian Province, Fujian Medical University, Fuzhou 350025, P. R. China;
3. Liver Disease Center, The First Affliated Hospital of Fujian Medical University, Fuzhou 350005, P. R. China;
4. The Key Lab of Analysis and Detection Technology for Food Safety of the MOE, Fujian Provincial Key Laboratory of Analysis and Detection Technology for Food Safety, College of Chemistry, Fuzhou University, Fuzhou 350002, P.R. China;
5. Center for Molecular Imaging and Translational Medicine, Xiamen University, Xiamen, 361005, P. R. China.
Abstract
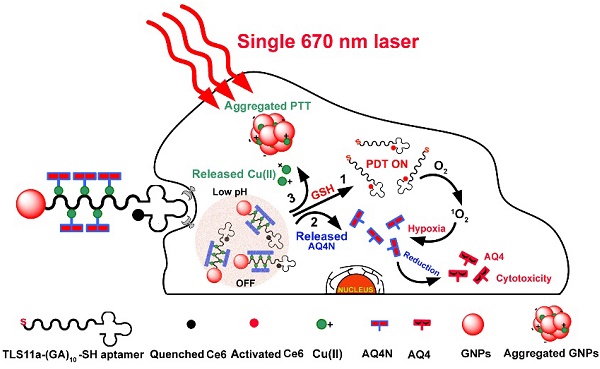
This study describes smart Cu(II)-aptamer complexes based gold nanoplatform for tumor micro-environment triggered programmable prodrug release, in demand photodynamic therapy and aggregation induced photothermal ablation of hepatocellular carcinoma. The nanoplatform is consist of monodispersed gold nanoparticle (GNP) that is binding to HCC cell specific targeting aptamers (TLS11a) through Au-S bond; the aptamer is labeled with Ce6 at the 5'end and coordinated with Cu(II) through (GA)10 repeating bases to load AQ4N at the 3' end. In normal physiological conditions, the fluorescence and ROS generation ability of Ce6 are quenched by GNPs via RET; but in cancerous cells, the fluorescence and the ROS generation of Ce6 could be recovered by cleavage of Au-S bond through high level of intracellular GSH for real-time imaging and in demand PDT. Meanwhile, the prodrug AQ4N release could be triggered by acid-cleavage of coordination bonds, then accompanied by a release of Cu(II) that would induce the electrostatic aggregation of GNPs for photo-thermal ablation; furthermore, the significantly enhanced chemotherapy efficiency could be achieved by PDT produced hypoxia to convert AQ4N into AQ4. In summary, here described nanoplatform with tumor cell specific responsive properties and programmable PDT/PTT/chemotherapy functions, might be an interesting synergistic strategy for HCC treatment.
Keywords: programmed synergistic therapy, Cu(II)-aptamer complexes, hypoxia induced chemotherapy, tumor micro-environment response, PDT and aggregation induced PTT.
 Global reach, higher impact
Global reach, higher impact