Impact Factor
Theranostics 2017; 7(1):40-50. doi:10.7150/thno.16676 This issue Cite
Review
Preclinical Applications of 3'-Deoxy-3'-[18F]Fluorothymidine in Oncology - A Systematic Review
1. European Institute for Molecular Imaging (EIMI), Westfälische Wilhelms-Universität (WWU) Münster, Münster, Germany.
2. Comprehensive Cancer Imaging Centre, Imperial College London, UK.
3. European Organization for Research and Treatment of Cancer Headquarters, Brussels, Belgium.
4. Department of Radiology and Nuclear Medicine, VU University Medical Center, Amsterdam, The Netherlands.
5. Imaging Sciences, University of Manchester, Manchester, UK.
6. Department of Oncology, Karmanos Cancer Institute, Wayne State University, Detroit, Michigan, USA.
7. Cancer Research UK Cambridge Institute, University of Cambridge, UK.
8. Department of Geriatric Medicine, Johanniter Hospital, Bonn, Germany.
* Equal contribution.
Abstract
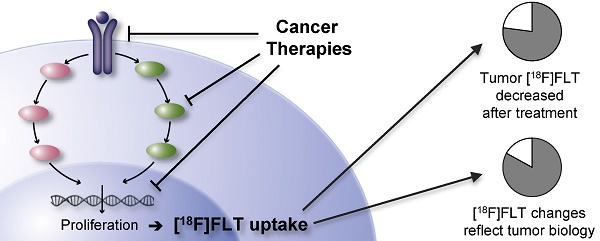
The positron emission tomography (PET) tracer 3'-deoxy-3'-[18F]fluorothymidine ([18F]FLT) has been proposed to measure cell proliferation non-invasively in vivo. Hence, it should provide valuable information for response assessment to tumor therapies. To date, [18F]FLT uptake has found limited use as a response biomarker in clinical trials in part because a better understanding is needed of the determinants of [18F]FLT uptake and therapy-induced changes of its retention in the tumor. In this systematic review of preclinical [18F]FLT studies, comprising 174 reports, we identify the factors governing [18F]FLT uptake in tumors, among which thymidine kinase 1 plays a primary role. The majority of publications (83 %) report that decreased [18F]FLT uptake reflects the effects of anticancer therapies. 144 times [18F]FLT uptake was related to changes in proliferation as determined by ex vivo analyses. Of these approaches, 77 % describe a positive relation, implying a good concordance of tracer accumulation and tumor biology. These preclinical data indicate that [18F]FLT uptake holds promise as an imaging biomarker for response assessment in clinical studies. Understanding of the parameters which influence cellular [18F]FLT uptake and retention as well as the mechanism of changes induced by therapy is essential for successful implementation of this PET tracer. Hence, our systematic review provides the background for the use of [18F]FLT in future clinical studies.
Keywords: positron emission tomography, FLT, Oncology.
 Global reach, higher impact
Global reach, higher impact