13.3
Impact Factor
Theranostics 2016; 6(12):2225-2234. doi:10.7150/thno.16260 This issue Cite
Research Paper
In Vitro and In Vivo Evaluation of 89Zr-DS-8273a as a Theranostic for Anti-Death Receptor 5 Therapy
1. Tumour Targeting Laboratory, Olivia Newton-John Cancer Research Institute, Melbourne, Australia;
2. School of Cancer Medicine, La Trobe University, Melbourne, Australia;
3. Department of Medicine, University of Melbourne, Melbourne, Australia;
4. Department of Medical Oncology, Austin Health, Heidelberg, Melbourne, Australia;
5. Department of Molecular Imaging and Therapy, Austin Health, Melbourne, Australia;
6. School of Engineering and Mathematical Sciences, La Trobe University, Melbourne, Australia;
7. School of Chemistry and Bio21 Molecular Science & Biotechnology Institute, University of Melbourne, Melbourne, Australia.
8. Translational Medicine & Clinical Pharmacology Department, Daiichi Sankyo Co Ltd, Tokyo, Japan;
9. Biologics Pharmacology Research Laboratories, Daiichi Sankyo Co Ltd, Tokyo, Japan.
10. Department of Translational Medicine and Clinical Pharmacology, Daiichi Sankyo Pharma Development, Edison, NJ, USA.
#Authors contributed equally to this work.
Abstract
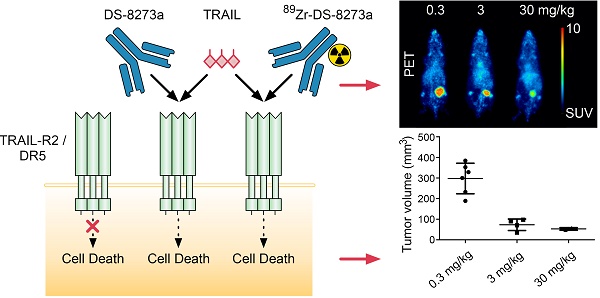
Background: DS-8273a, an anti-human death receptor 5 (DR5) agonistic antibody, has cytotoxic activity against human cancer cells and induces apoptosis after specific binding to DR5. DS-8273a is currently being used in clinical Phase I trials. This study evaluated the molecular imaging of DR5 expression in vivo in mouse tumor models using SPECT/CT and PET/MRI, as a tool for drug development and trial design. Methods: DS-8273a was radiolabeled with indium-111 and zirconium-89. Radiochemical purity, immunoreactivity, antigen binding affinity and serum stability were assessed in vitro. In vivo biodistribution and pharmacokinetic studies were performed, including SPECT/CT and PET/MR imaging. A dose-escalation study using a PET/MR imaging quantitative analysis was also performed to determine DR5 receptor saturability in a mouse model. Results: 111In-CHX-A″-DTPA-DS-8273a and 89Zr-Df-Bz-NCS-DS-8273a showed high immunoreactivity (100%), high serum stability, and bound to DR5 expressing cells with high affinity (Ka, 1.02-1.22 × 1010 M-1). The number of antibodies bound per cell was 32,000. In vivo biodistribution studies showed high and specific uptake of 111In-CHX-A″-DTPA-DS-8273a and 89Zr-Df-Bz-NCS-DS-8273a in DR5 expressing COLO205 xenografts, with no specific uptake in normal tissues or in DR5-negative CT26 xenografts. DR5 receptor saturation was observed in vivo by biodistribution studies and quantitative PET/MRI analysis. Conclusion: 89Zr-Df-Bz-NCS-DS-8273a is a potential novel PET imaging reagent for human bioimaging trials, and can be used for effective dose assessment and patient response evaluation in clinical trials.
Keywords: DS-8273a, death receptor 5, apoptosis, zirconium-89, colorectal cancer, PET/MRI.
 Global reach, higher impact
Global reach, higher impact