13.3
Impact Factor
Theranostics 2016; 6(12):2068-2083. doi:10.7150/thno.15007 This issue Cite
Research Paper
Crucial Role of miR-433 in Regulating Cardiac Fibrosis
1. Department of Cardiology, The First Affiliated Hospital of Nanjing Medical University, Nanjing 210029, China.
2. Cardiac Regeneration and Ageing Lab, School of Life Science, Shanghai University, Shanghai 200444, China.
3. Laboratory of Experimental Cardiology, University Medical Centre Utrecht, Utrecht 3508GA, The Netherlands.
4. Department of Cardiology, Tongji Hospital, Tongji University School of Medicine, Shanghai 200065, China.
5. Cardiovascular Research Center and Department of Physiology, Temple University School of Medicine, Philadelphia, PA 19140, USA.
6. Cardiovascular Division of the Massachusetts General Hospital and Harvard Medical School, Boston, MA 02215, USA.
7. Victor Babes National Institute of Pathology, Bucharest 050096, Romania.
8. Division of Cellular and Molecular Biology and Histology, Carol Davila University of Medicine and Pharmacy, Bucharest 050474, Romania.
9. Innovative Drug Research Center of Shanghai University, Shanghai 200444, China.
10. State Key Laboratory of Medical Genomics & Shanghai Institute of Hypertension, Ruijin Hospital Affiliated to Shanghai Jiao Tong University School of Medicine, Shanghai 200025, China.
* These two authors contributed equally to this work.
Abstract
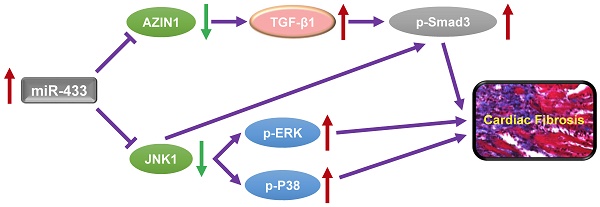
Dysregulation of microRNAs has been implicated in many cardiovascular diseases including fibrosis. Here we report that miR-433 was consistently elevated in three models of heart disease with prominent cardiac fibrosis, and was enriched in fibroblasts compared to cardiomyocytes. Forced expression of miR-433 in neonatal rat cardiac fibroblasts increased proliferation and their differentiation into myofibroblasts as determined by EdU incorporation, α-SMA staining, and expression levels of fibrosis-associated genes. Conversely, inhibition of miR-433 exhibited opposite results. AZIN1 and JNK1 were identified as two target genes of miR-433. Decreased level of AZIN1 activated TGF-β1 while down-regulation of JNK1 resulted in activation of ERK and p38 kinase leading to Smad3 activation and ultimately cardiac fibrosis. Importantly, systemic neutralization of miR-433 or adeno-associated virus 9 (AAV9)-mediated cardiac transfer of a miR-433 sponge attenuated cardiac fibrosis and ventricular dysfunction following myocardial infarction. Thus, our work suggests that miR-433 is a potential target for amelioration of cardiac fibrosis.
Keywords: cardiac fibrosis, miR-433, AZIN1, JNK1.
 Global reach, higher impact
Global reach, higher impact