13.3
Impact Factor
Theranostics 2016; 6(11):1877-1886. doi:10.7150/thno.15284 This issue Cite
Research Paper
Aptamer-Mediated Transparent-Biocompatible Nanostructured Surfaces for Hepatocellular Circulating Tumor Cells Enrichment
1. Department of Oncology, Zhongnan Hospital of Wuhan University; Hubei Key Laboratory of Tumor Biological Behaviors & Hubei Cancer Clinical Study Center, Wuhan 430071, P. R. China;
2. Department of Oncology, Peking University Shenzhen Hospital, No. 1120, Lianhua Road, Futain District, Shenzhen 518036, Guangdong Province, P. R. China;
3. Department of Breast Cancer, Cancer Center, Guangdong General Hospital, Guangzhou 510050, Guangdong Province, P.R. China.
*These authors contribute to this study equally.
Abstract
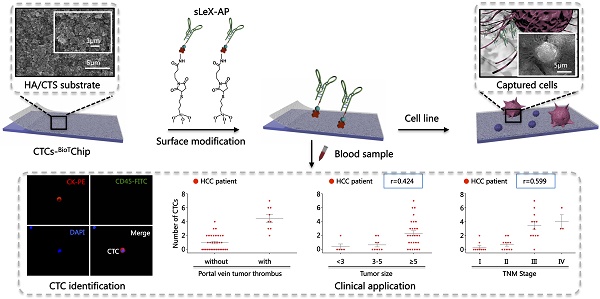
Circulating tumor cells (CTCs) have been considered as the origin of cancer metastasis. Thus, detection of CTCs in peripheral blood is of great value in different types of solid tumors. However, owing to extremely low abundance of CTCs, detection of them has been technically challenging. To establish a simple and efficient method for CTCs detection in patients with hepatocellular carcinoma (HCC), we applied biocompatible and transparent HA/CTS (Hydroxyapatite/chitosan) nanofilm to achieve enhanced topographic interactions with nanoscale cellular surface components, and we used sLex-AP (aptamer for carbohydrate sialyl Lewis X) to coat onto HA/CTS nanofilm for efficient capture of HCC CTCs, these two functional components combined to form our CTC-BioTChip platform. Using this platform, we realized HCC CTCs' capture and identification, the average recovery rate was 61.6% or more at each spiking level. Importantly, our platform identified CTCs (2±2 per 2 mL) in 25 of 42 (59.5%) HCC patients. Moreover, both the positivity rate and the number of detected CTCs were significantly correlated with tumor size, portal vein tumor thrombus, and the TNM (tumor-node-metastasis) stage. In summary, our CTC-BioTChip platform provides a new method allowing for simple but efficient detection of CTCs in HCC patients, and it holds potential of clinically usefulness in monitoring HCC prognosis and guiding individualized treatment in the future.
Keywords: Circulating tumor cells, Cell capture, Aptamer, Hepatocellular carcinoma, Nanomaterial, Hydroxyapatite/chitosan.
 Global reach, higher impact
Global reach, higher impact