13.3
Impact Factor
Theranostics 2016; 6(11):1768-1779. doi:10.7150/thno.15929 This issue Cite
Research Paper
Targeting Amino Acid Metabolism for Molecular Imaging of Inflammation Early After Myocardial Infarction
1. Department of Nuclear Medicine, Hannover Medical School, Hannover, Germany;
2. Division of Molecular and Translational Cardiology, Department of Cardiology and Angiology, Hannover Medical School, Hannover, Germany.
Abstract
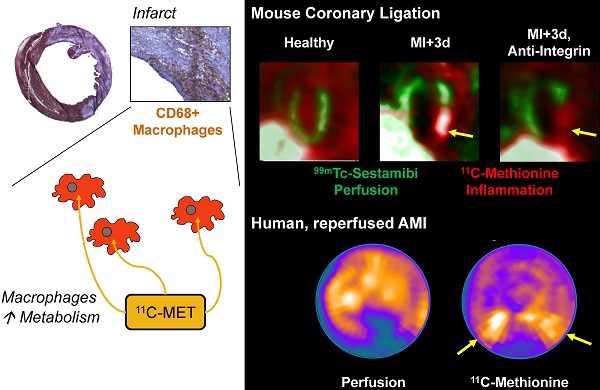
Acute tissue inflammation after myocardial infarction influences healing and remodeling and has been identified as a target for novel therapies. Molecular imaging holds promise for guidance of such therapies. The amino acid 11C-methionine is a clinically approved agent which is thought to accumulate in macrophages, but not in healthy myocytes. We assessed the suitability of positron emission tomography (PET) with 11C-methionine for imaging post-MI inflammation, from cell to mouse to man. Uptake assays demonstrated 7-fold higher 11C-methionine uptake by polarized pro-inflammatory M1 macrophages over anti-inflammatory M2 subtypes (p<0.001). C57Bl/6 mice (n=27) underwent coronary artery ligation or no surgery. Serial 11C-methionine PET was performed 3, 5 and 7d later. MI mice exhibited a perfusion defect in 32-50% of the left ventricle (LV). PET detected increased 11C-methionine accumulation in the infarct territory at 3d (5.9±0.9%ID/g vs 4.7±0.9 in remote myocardium, and 2.6±0.5 in healthy mice; p<0.05 and <0.01 respectively), which declined by d7 post-MI (4.3±0.6 in infarct, 3.4±0.8 in remote; p=0.03 vs 3d, p=0.08 vs healthy). Increased 11C-methionine uptake was associated with macrophage infiltration of damaged myocardium. Treatment with anti-integrin antibodies (anti-CD11a, -CD11b, -CD49d; 100µg) lowered macrophage content by 56% and 11C-methionine uptake by 46% at 3d post-MI. A patient study at 3d after ST-elevation MI and early reperfusion confirmed elevated 11C-methionine uptake in the hypoperfused myocardial region. Targeting of elevated amino acid metabolism in pro-inflammatory M1 macrophages enables PET imaging-derived demarcation of tissue inflammation after MI. 11C-methionine-based molecular imaging may assist in the translation of novel image-guided, inflammation-targeted regenerative therapies.
Keywords: inflammation, myocardial infarction, 11C-methionine, macrophages, positron emission tomography (PET).
 Global reach, higher impact
Global reach, higher impact