13.3
Impact Factor
Theranostics 2015; 5(12):1317-1327. doi:10.7150/thno.12494 This issue Cite
Research Paper
Blood Accessibility to Fibrin in Venous Thrombosis is Thrombus Age-Dependent and Predicts Fibrinolytic Efficacy: An In Vivo Fibrin Molecular Imaging Study
1. Cardiovascular Research Center, Division of Cardiology, Massachusetts General Hospital, Harvard Medical School
2. Center for Systems Biology, Massachusetts General Hospital, Harvard Medical School
3. Wellman Center for Photomedicine, Massachusetts General Hospital, Harvard Medical School
4. Department of Medicine, University of Tennessee Health Sciences Center
5. Section of Vascular Surgery, Department of Surgery, University of Michigan, Ann Arbor, MI
6. Current Address: Istanbul Medipol University
*AFS and CWK contributed equally
† FAJ and JRM share senior authorship
Abstract
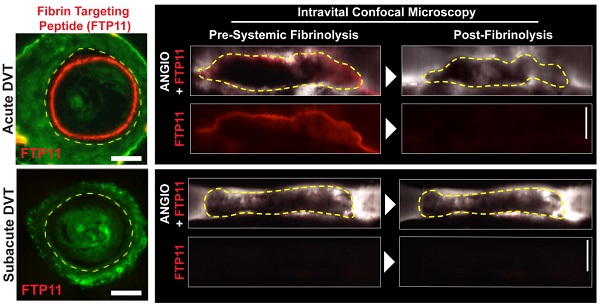
Fibrinolytic therapy of venous thromboembolism (VTE) is increasingly utilized, yet limited knowledge is available regarding in vivo mechanisms that govern fibrinolytic efficacy. In particular, it is unknown how age-dependent thrombus organization limits direct blood contact with fibrin, the target of blood-based fibrinolytic agents. Utilizing high-resolution in vivo optical molecular imaging with FTP11, a near-infrared fluorescence (NIRF) fibrin-specific reporter, here we investigated the in vivo interrelationships of blood accessibility to fibrin, thrombus age, thrombus neoendothelialization, and fibrinolysis in murine venous thrombosis (VT). In both stasis VT and non-stasis VT, NIRF microscopy showed that FTP11 fibrin binding was thrombus age-dependent. FTP11 localized to the luminal surface of early-stage VT, but only minimally to subacute VT (p<0.001). Transmission electron microscopy of early stage VT revealed direct blood cell contact with luminal fibrin-rich surfaces. In contrast, subacute VT exhibited an encasing CD31+ neoendothelial layer that limited blood cell contact with thrombus fibrin in both VT models. Next we developed a theranostic strategy to predict fibrinolytic efficacy based on the in vivo fibrin accessibility to blood NIRF signal. Mice with variably aged VT underwent FTP11 injection and intravital microscopy (IVM), followed by tissue plasminogen activator infusion to induce VT fibrinolysis. Fibrin molecular IVM revealed that early stage VT, but not subacute VT, bound FTP11 (p<0.05), and experienced higher rates of fibrinolysis and total fibrinolysis (p<0.05 vs. subacute VT). Before fibrinolysis, the baseline FTP11 NIRF signal predicted the net fibrinolysis at 60 minutes (p<0.001). Taken together, these data provide novel insights into the temporal evolution of VT and its susceptibility to therapeutic fibrinolysis. Fibrin molecular imaging may provide a theranostic strategy to identify venous thrombi amenable to fibrinolytic therapies.
Keywords: Fibrin, fibrinolysis, venous thrombosis, molecular imaging, pulmonary embolism
 Global reach, higher impact
Global reach, higher impact