13.3
Impact Factor
Theranostics 2015; 5(12):1303-1316. doi:10.7150/thno.12956 This issue Cite
Research Paper
First-in-human uPAR PET: Imaging of Cancer Aggressiveness
1. Department of Clinical Physiology, Nuclear Medicine & PET and Cluster for Molecular Imaging, Rigshospitalet and University of Copenhagen, Denmark.
2. Department of Physiology and Nuclear Medicine, University hospital of Copenhagen, Gentofte, Denmark
3. Finsen Laboratory, Rigshospitalet, Biotech Research & Innovation Centre, University of Copenhagen, Copenhagen, Denmark
4. Department of Oncology, Finsen Centre, Rigshospitalet, Copenhagen, Denmark.
5. Copenhagen Prostate Cancer Center, Department of Urology, Abdominal Centre, Rigshospitalet, Copenhagen, Denmark.
6. Department of Plastic Surgery and Burns Treatment, Rigshospitalet, Copenhagen, Denmark.
# These authors contributed equally to this work.
Abstract
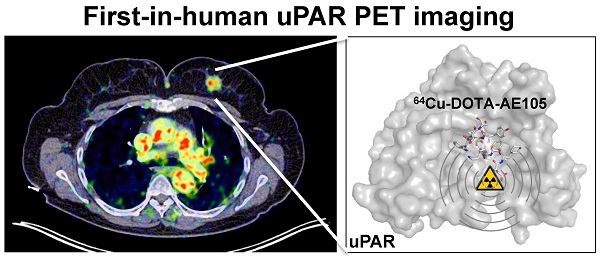
A first-in-human clinical trial with Positron Emission Tomography (PET) imaging of the urokinase-type plasminogen activator receptor (uPAR) in patients with breast, prostate and bladder cancer, is described. uPAR is expressed in many types of human cancers and the expression is predictive of invasion, metastasis and indicates poor prognosis. uPAR PET imaging therefore holds promise to be a new and innovative method for improved cancer diagnosis, staging and individual risk stratification. The uPAR specific peptide AE105 was conjugated to the macrocyclic chelator DOTA and labeled with 64Cu for targeted molecular imaging with PET. The safety, pharmacokinetic, biodistribution profile and radiation dosimetry after a single intravenous dose of 64Cu-DOTA-AE105 were assessed by serial PET and computed tomography (CT) in 4 prostate, 3 breast and 3 bladder cancer patients. Safety assessment with laboratory blood screening tests was performed before and after PET ligand injection. In a subgroup of the patients, the in vivo stability of our targeted PET ligand was determined in collected blood and urine. No adverse or clinically detectable side effects in any of the 10 patients were found. The ligand exhibited good in vivo stability and fast clearance from plasma and tissue compartments by renal excretion. In addition, high uptake in both primary tumor lesions and lymph node metastases was seen and paralleled high uPAR expression in excised tumor tissue. Overall, this first-in-human study therefore provides promising evidence for safe use of 64Cu-DOTA-AE105 for uPAR PET imaging in cancer patients.
Keywords: clinical trial, PET, uPAR
 Global reach, higher impact
Global reach, higher impact