13.3
Impact Factor
Theranostics 2019; 9(18):5298-5314. doi:10.7150/thno.34559 This issue Cite
Research Paper
LncRNA-MTA2TR functions as a promoter in pancreatic cancer via driving deacetylation-dependent accumulation of HIF-1α
1. Department of Emergency Surgery, Union Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan 430022, China.
2. Department of Pancreatic Surgery, Union Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan 430022, China.
# These authors contributed equally to this work.
Abstract
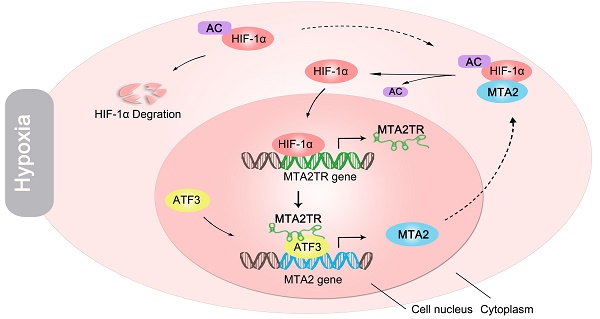
Rationale: Hypoxia has been proved to contribute to aggressive phenotype of cancers, while functional and regulatory mechanism of long noncoding RNA (lncRNA) in the contribution of hypoxia on pancreatic cancer (PC) tumorigenesis is incompletely understood. The aim of this study was to uncover the regulatory and functional roles for hypoxia-induced lncRNA-MTA2TR (MTA2 transcriptional regulator RNA, AF083120.1) in the regulation of PC tumorigenesis.
Methods: A lncRNA microarray confirmed MTA2TR expression in tissues of PC patients. The effects of MTA2TR on proliferation and metastasis of PC cells and xenograft models were determined, and the key mechanisms by which MTA2TR promotes PC were further dissected. Furthermore, the expression and regulation of MTA2TR under hypoxic conditions in PC cells were assessed. We also assessed the correlation between MTA2TR expression and PC patient clinical outcomes.
Results: We found that metastasis associated protein 2 (MTA2) transcriptional regulator lncRNA (MTA2TR) was overexpressed in PC patient tissues relative to paired noncancerous tissues. Furthermore, we found that depletion of MTA2TR significantly inhibited PC cell proliferation and invasion both in vitro and in vivo. We further demonstrated that MTA2TR transcriptionally upregulates MTA2 expression by recruiting activating transcription factor 3 (ATF3) to the promoter area of MTA2. Consequentially, MTA2 can stabilize the HIF-1α protein via deacetylation, which further activates HIF-1α transcriptional activity. Interestingly, our results revealed that MTA2TR is transcriptionally regulated by HIF-1α under hypoxic conditions. Our clinical samples further indicated that the overexpression of MTA2TR was correlated with MTA2 upregulation, as well as with reduced overall survival (OS) in PC patients.
Conclusions: These results suggest that feedback between MTA2TR and HIF-1α may play a key role in regulating PC tumorigenesis, thus potentially highlighting novel avenues PC treatment.
Keywords: pancreatic cancer, LncRNA-MTA2TR, MTA2, ATF3, HIF-1α
 Global reach, higher impact
Global reach, higher impact