13.3
Impact Factor
Theranostics 2019; 9(15):4558-4566. doi:10.7150/thno.31052 This issue Cite
Research Paper
LncRNA DCRF regulates cardiomyocyte autophagy by targeting miR-551b-5p in diabetic cardiomyopathy
1. Department of Endocrinology, The Second Affiliated Hospital of Soochow University, Suzhou, China
2. Jiangsu Key Laboratory of Neuropsychiatric Diseases and Institute of Neuroscience, Soochow University, Suzhou, China
3. Department of Cardiology, The Second Affiliated Hospital of Soochow University, Suzhou, China
# These authors contributed equally to this work.
Abstract
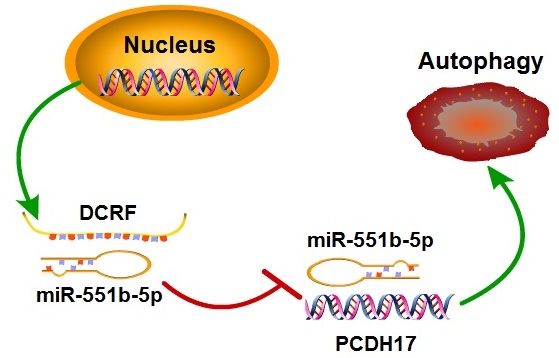
Background: We generated a rat model of diabetic cardiomyopathy (DCM) and reported significant upregulation of the long non-coding RNA DCRF. This study was designed to determine the molecular mechanisms of DCRF in the development of DCM.
Methods: Real-time PCR and RNA fluorescent in situ hybridization were conducted to detect the expression pattern of DCRF in cardiomyocytes. Histological and echocardiographic analyses were used to assess the effect of DCRF knockdown on cardiac structure and function in diabetic rats. mRFP-GFP-LC3 fluorescence microscopy, transmission electron microscopy, and Western blotting were carried out to determine cardiomyocyte autophagy. RNA immunoprecipitation and luciferase reporter assays were performed to elucidate the regulatory role of DCRF/miR-551b-5p/PCDH17 pathway in cardiomyocyte autophagy.
Results: Our findings showed that DCRF knockdown reduced cardiomyocyte autophagy, attenuated myocardial fibrosis, and improved cardiac function in diabetic rats. High glucose increased DCRF expression and induced autophagy in cardiomyocytes. RNA immunoprecipitation and luciferase reporter assays indicated that DCRF was targeted by miR-551b-5p in an AGO2-dependent manner and PCDH17 was the direct target of miR-551b-5p. Forced expression of DCRF was found to attenuate the inhibitory effect of miR-551b-5p on PCDH17. Furthermore, DCRF knockdown decreased PCDH17 expression and suppressed autophagy in cardiomyocytes treated with high glucose.
Conclusion: Our study suggests that DCRF can act as a competing endogenous RNA to increase PCDH17 expression by sponging miR-551b-5p, thus contributing to increased cardiomyocyte autophagy in DCM.
Keywords: autophagy, DCRF, diabetic cardiomyopathy, miR-551b-5p
 Global reach, higher impact
Global reach, higher impact