13.3
Impact Factor
Theranostics 2019; 9(14):4192-4207. doi:10.7150/thno.34157 This issue Cite
Research Paper
PMMA-Fe3O4 for internal mechanical support and magnetic thermal ablation of bone tumors
1. Department of Orthopaedics, Second Affiliated Hospital of Chongqing Medical University, 76 Linjiang Road, Yuzhong Distinct, Chongqing, 400010, P. R. China.
2. Chongqing Key Laboratory of Ultrasound Molecular Imaging, Second Affiliated Hospital of Chongqing Medical University, 76 Linjiang Road, Yuzhong Distinct, Chongqing, 400010, P. R. China.
3. Shanghai Institute of Ultrasound in Medicine, Shanghai Jiao Tong University Affiliated Sixth People's Hospital, 600 Yishan Road, Xuhui Distinct, Shanghai, 200233, P. R. China.
4. Radiology department of University Hospital, Case Western Reserve University, 10900 Euclid Ave, Cleveland, OH, 44106, USA.
5. Department of Physics, Ryerson University, Toronto, 350 Victoria Street Toronto, Ontario M5B 2K3, Ontario, Canada
6. Guangdong Provincial Key Laboratory of Robotics and Intelligent System, Shenzhen Institutes of Advanced Technology, Chinese Academy of Sciences, Shenzhen 518055, China
7. Department of Radiology, Second Affiliated Hospital of Chongqing Medical University, 76 Linjiang Road, Yuzhong Distinct, Chongqing, 400010, P. R. China.
# Kexiao Yu and Bing Liang are co-first authors who contributed equally to this study.
Abstract
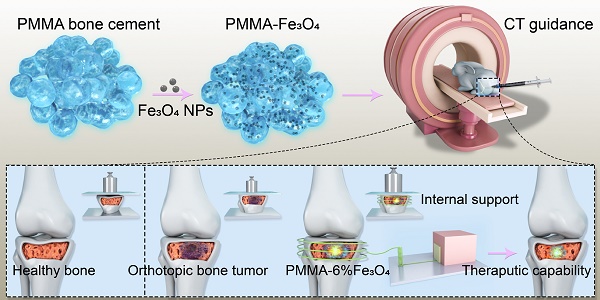
Background: Minimally invasive modalities are of great interest in the field of treating bone tumors. However, providing reliable mechanical support and fast killing of tumor cells to achieve rapid recovery of physical function is still challenging in clinical works.
Methods: A material with two functions, mechanical support and magnetic thermal ablation, was developed from Fe3O4 nanoparticles (NPs) distributed in a polymethylmethacrylate (PMMA) bone cement. The mechanical properties and efficiency of magnetic field-induced thermal ablation were systematically and successfully evaluated in vitro and ex vivo. CT images and pathological examination were successfully applied to evaluate therapeutic efficacy with a rabbit bone tumor model. Biosafety evaluation was performed with a rabbit in vivo, and a cytotoxicity test was performed in vitro.
Results: An NP content of 6% Fe3O4 (PMMA-6% Fe3O4, mFe: 0.01 g) gave the most suitable performance for in vivo study. At the 56-day follow-up after treatment, bone tumors were ablated without obvious side effects. The pathological examination and new bone formation in CT images clearly illustrate that the bone tumors were completely eliminated. Correspondingly, after treatment, the tendency of bone tumors toward metastasis significantly decreased. Moreover, with well-designed mechanical properties, PMMA-6%Fe3O4 implantation endowed tumor-bearing rabbit legs with excellent bio-mimic bone structure and internal support. Biosafety evaluation did not induce an increase or decrease in the immune response, and major functional parameters were all at normal levels.
Conclusion: We have presented a novel, highly efficient and minimally invasive approach for complete bone tumor regression and bone defect repair by magnetic thermal ablation based on PMMA containing Fe3O4 NPs; this approach shows excellent heating ability for rabbit VX2 tibial plateau tumor ablation upon exposure to an alternating magnetic field (AMF) and provides mechanical support for bone repair. The new and powerful dual-function implant is a promising minimally invasive agent for the treatment of bone tumors and has good clinical translation potential.
Keywords: magnetic thermal ablation, PMMA, bone tumors, mechanical support, fast clinical translation
 Global reach, higher impact
Global reach, higher impact