13.3
Impact Factor
Theranostics 2019; 9(9):2526-2540. doi:10.7150/thno.32915 This issue Cite
Research Paper
Metastatic Colorectal Cancer Rewrites Metabolic Program Through a Glut3-YAP-dependent Signaling Circuit
1. Department of Biochemistry and Molecular Cell Biology, School of Medicine, College of Medicine, Taipei Medical University, Taipei, Taiwan.
2. Graduate Institute of Medical Sciences, College of Medicine, Taipei Medical University, Taipei, Taiwan.
3. Department of Cytopathology, Chi Mei Medical Center, Tainan, Taiwan.
4. TMU Research Center of Cancer Translational Medicine, Taipei Medical University, Taipei, Taiwan.
5. Center for Cell Therapy and Regeneration Medicine, Taipei Medical University, Taipei, Taiwan.
*Chih-Chia Kuo, Hsiang-Hsi Ling, and Ming-Chen Chiang contributed equally to this work
Abstract
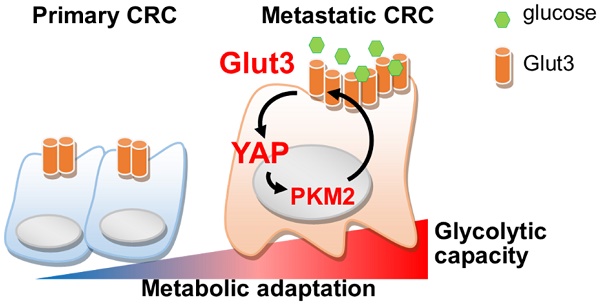
Rationale: Cancer cells reprogram cellular metabolism to fulfill their needs for rapid growth and metastasis. However, the mechanism controlling this reprogramming is poorly understood. We searched for upregulated signaling in metastatic colorectal cancer and investigated the mechanism by which Glut3 promotes tumor metastasis.
Methods: We compared RNA levels and glycolytic capacity in primary and metastatic colon cancer. The expression and association of Glut3 with clinical prognosis in colon cancer tissues was determined by immunohistochemistry. Glut3 gain-of-function and loss-of-function were established using colon cancer HCT116, HT29, and metastatic 116-LM cells, and tumor invasiveness and stemness properties were evaluated. Metabolomic profiles were analyzed by GC/MS and CE-TOF/MS. The metastatic burden in mice fed a high-fat sucrose diet was assessed by intravenous inoculation with Glut3 knockdown 116-LM cells.
Results: Upregulation of glycolytic genes and glycolytic capacity was detected in metastatic colorectal cancer cells. Specifically, Glut3 overexpression was associated with metastasis and poor survival in colorectal cancer patients. Mechanistically, Glut3 promoted invasiveness and stemness in a Yes-associated protein (YAP)-dependent manner. Activation of YAP in turn transactivated Glut3 and regulated a group of glycolytic genes. Interestingly, the expression and phosphorylation of PKM2 were concomitantly upregulated in metastatic colorectal cancer, and it was found to interact with YAP and enhance the expression of Glut3. Importantly, a high-fat high-sucrose diet promoted tumor metastasis, whereas the inhibition of either Glut3 or YAP effectively reduced the metastatic burden.
Conclusion: Activation of the Glut3-YAP signaling pathway acts as a master activator to reprogram cancer metabolism and thereby promotes metastasis. Our findings reveal the importance of metabolic reprogramming in supporting cancer metastasis as well as possible therapeutic targets.
Keywords: cancer metabolism, colorectal cancer, glucose transporter, metastasis, YAP
 Global reach, higher impact
Global reach, higher impact