13.3
Impact Factor
Theranostics 2019; 9(2):436-448. doi:10.7150/thno.27576 This issue Cite
Research Paper
MSC-Delivered Soluble TRAIL and Paclitaxel as Novel Combinatory Treatment for Pancreatic Adenocarcinoma
1. Division of Oncology, Department of Medical and Surgical Sciences for Children & Adults, University-Hospital of Modena and Reggio Emilia, Modena, Italy;
2. Rigenerand srl, Medolla, Modena, Italy;
3. International Centre for Genetic Engineering and Biotechnology, Trieste, Italy;
4. Technopole of Mirandola TPM, Mirandola, Modena;
5. Department of Life Sciences, University of Modena and Reggio Emilia, Modena, Italy;
6. Department of Diagnostic and Clinical Medicine and of Public Health, Institute of Pathology, University of Modena and Reggio Emilia, Modena, Italy;
7. Pharmacy Unit, University-Hospital of Modena and Reggio Emilia, Modena, Italy;
8. Division of Plastic and Reconstructive Surgery, University-Hospital of Modena and Reggio Emilia, Modena, Italy;
9. Aflac Cancer and Blood Disorders Center, Children's Healthcare of Atlanta, Atlanta, GA, USA;
10. Department of Pediatrics, Emory University, Atlanta, GA, USA;
11. Department of Surgery, Oncology and Gastroenterology, University of Padua, Padua, Italy;
12. Medical Oncology 2, Veneto Institute of Oncology IRCCS, Padua, Italy.
Abstract
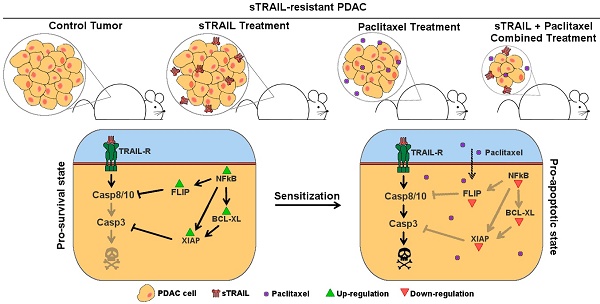
Pancreatic cancer is the fourth leading cause of cancer death in western countries with more than 100,000 new cases per year in Europe and a mortality rate higher than 90%. In this scenario, advanced therapies based on gene therapies are emerging, thanks to a better understanding of tumour architecture and cancer cell alterations. We have demonstrated the efficacy of an innovative approach for pancreatic cancer based on mesenchymal stromal cells (MSC) genetically engineered to produce TNF-related Apoptosis Inducing Ligand (TRAIL). Here we investigated the combination of this MSC-based approach with the administration of a paclitaxel (PTX)-based chemotherapy to improve the potential of the treatment, also accounting for a possible resistance onset.
Methods: Starting from the BXPC3 cell line, we generated and profiled a TRAIL-resistant model of pancreatic cancer, testing the impact of the combined treatment in vitro with specific cytotoxicity and metabolic assays. We then challenged the rationale in a subcutaneous mouse model of pancreatic cancer, assessing its effect on tumour size accounting stromal and parenchymal organization.
Results: PTX was able to restore pancreatic cancer sensitivity to MSC-delivered TRAIL by reverting its pro-survival gene expression profile. The two compounds cooperate both in vitro and in vivo and the combined treatment resulted in an improved cytotoxicity on tumour cells.
Conclusion: In summary, this study uncovers the potential of a combinatory approach between MSC-delivered TRAIL and PTX, supporting the combination of cell-based products and conventional chemotherapeutics as a tool to improve the efficacy of the treatments, also addressing possible mechanisms of resistance.
Keywords: pancreatic cancer, combinatory, synergy, TRAIL, paclitaxel
 Global reach, higher impact
Global reach, higher impact