13.3
Impact Factor
Theranostics 2018; 8(22):6291-6306. doi:10.7150/thno.28845 This issue Cite
Research Paper
Rational design of temperature-sensitive blood-vessel-embolic nanogels for improving hypoxic tumor microenvironment after transcatheter arterial embolization
1. National Engineering Research Center for Nanomedicine, College of Life Science and Technology, Huazhong University of Science and Technology, 430074, Wuhan City, P. R. China.
2. Hubei Province Key Laboratory of Molecular Imaging, Department of Radiology, Union Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan 430022, China.
3. Shenzhen Institute of Huazhong University of Science and Technology, 518057, Shenzhen, P. R. China
* These authors contributed equally to this work.
Abstract
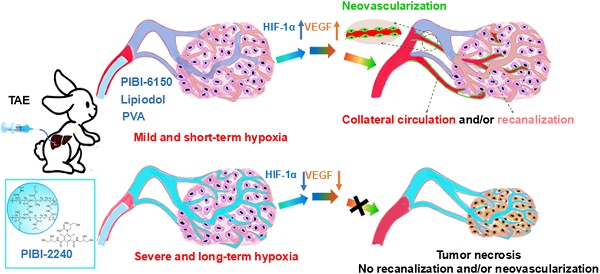
Transcatheter arterial embolization (TAE) plays an important role in clinical tumor therapy by accomplishing vessel-casting embolization of tumor arteries at all levels and suppressing tumor collateral circulation and vascular re-canalization. In this study, we describe smart blood-vessel-embolic nanogels for improving the anti-tumor efficacy of TAE therapy on hepatocellular carcinoma (HCC).
Methods: In this study, an in vitro model composed of two microfluidic chips was used for simulating the tumor capillary network and analyzing artery-embolization properties. Also, blood-vessel-casting embolization of renal arteries was evaluated in normal rabbits. Using a VX2 tumor-bearing rabbit model, the therapeutic efficacy of TAE on HCC was investigated for tumor growth, necrosis, and proliferation. Neovascularization and collateral circulation were evaluated by immunofluorescent detection of hypoxia-inducible factor-1α (HIF-1α), vascular endothelial growth factor (VEGF), and CD31 following the TAE therapy of VX2 tumor-bearing rabbits.
Results: Sufficient embolization of all eight levels of micro-channels was achieved in a tumor-vessel-mimetic model with two microfluidic chips using PIBI-2240, and was further confirmed in renal arteries of normal rabbit. Effective inhibition of tumor collateral circulation and vascular re-canalization was observed in VX2 tumor-bearing rabbits due to the reduced expression levels of HIF-1α, VEGF, and CD31.
Conclusions: The exceptional anti-tumor effect of PIBI-2240 observed in this study suggested that it is an excellent blood-vessel-embolic material for tumor TAE therapy.
Keywords: Temperature-sensitive, Nanogel, Transcatheter arterial embolization, hypoxic microenvironment
 Global reach, higher impact
Global reach, higher impact