Impact Factor
Theranostics 2018; 8(20):5529-5547. doi:10.7150/thno.29039 This issue Cite
Research Paper
Targeting Mitochondrial Dysfunction and Oxidative Stress in Activated Microglia using Dendrimer-Based Therapeutics
1. Center for Nanomedicine, Department of Ophthalmology, Wilmer Eye Institute Johns Hopkins University School of Medicine, Baltimore, MD 21231, USA;
2. Department of Chemical and Biomolecular Engineering, Johns Hopkins University, Baltimore MD, 21218, USA.
3. Department of Anesthesiology and Critical Care Medicine, Johns Hopkins University School of Medicine, Baltimore, MD 21287, USA.
4. Hugo W. Moser Research Institute at Kennedy Krieger, Inc., Baltimore MD, 21205, USA.
5. Kennedy Krieger Institute - Johns Hopkins University for Cerebral Palsy Research Excellence, Baltimore, MD 21218, USA.
*These authors contributed equally
Abstract
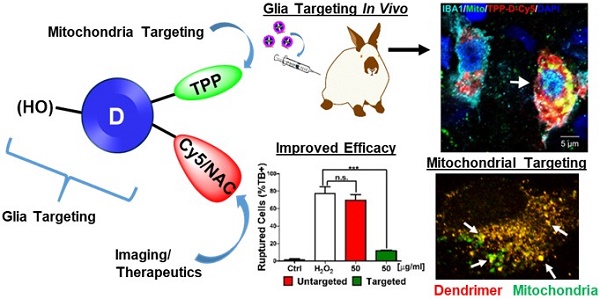
Mitochondrial oxidative stress is associated with many neurodegenerative diseases, such as traumatic brain injury (TBI). Targeted delivery of antioxidants to mitochondria has failed to translate into clinical success due to their nonspecific cellular localization, poor transport properties across multiple biological barriers, and associated side effects. These challenges, coupled with the complex function of the mitochondria, create the need for innovative delivery strategies.
Methods: Neutral hydroxyl-terminated polyamidoamine (PAMAM) dendrimers have shown significant potential as nanocarriers in multiple brain injury models. N-acetyl cysteine (NAC) is a clinically used antioxidant and anti-inflammatory agent which has shown significant potency when delivered in a targeted manner. Here we present a mitochondrial targeting hydroxyl PAMAM dendrimer-drug construct (TPP-D-NAC) with triphenyl-phosphonium (TPP) for mitochondrial targeting and NAC for targeted delivery to mitochondria in injured glia. Co-localization and mitochondrial content of mitochondria-targeted and unmodified dendrimer were assessed in microglia and macrophages in vitro via immunohistochemistry and fluorescence quantification. Therapeutic improvements of TPP-D-NAC over dendrimer-NAC conjugate (D-NAC) and free NAC were evaluated in vitro in microglia under oxidative stress challenge. In vivo neuroinflammation targeting was confirmed in a rabbit model of TBI.
Results: TPP-conjugated dendrimer co-localized significantly more with mitochondria than unmodified dendrimer without altering overall levels of cellular internalization. This targeting capability translated to significant improvements in the attenuation of oxidative stress by TPP-D-NAC compared to D-NAC and free NAC. Upon systemic administration in a rabbit TBI model, TPP-conjugated dendrimer co-localized specifically with mitochondria in activated microglia and macrophages in the white matter of the ipsilateral/injured hemisphere, confirming its BBB penetration and glial targeting capabilities.
Conclusion: D-NAC has shown promising efficacy in many animal models of neurodegeneration, and this work provides evidence that modification for mitochondrial targeting can further enhance its therapeutic efficacy, particularly in diseases where oxidative stress-induced glial cell death plays a significant role in disease progression.
Keywords: Hydroxyl PAMAM dendrimer, mitochondria, activated microglia, sub-cellular targeting, oxidative stress, N-acetyl cysteine
 Global reach, higher impact
Global reach, higher impact