13.3
Impact Factor
Theranostics 2018; 8(18):5106-5125. doi:10.7150/thno.26585 This issue Cite
Research Paper
Theranostic pretargeted radioimmunotherapy of internalizing solid tumor antigens in human tumor xenografts in mice: Curative treatment of HER2-positive breast carcinoma
1. Molecular Pharmacology Program, Memorial Sloan Kettering Cancer Center, New York, NY 10065, USA;
2. Department of Radiology, Memorial Sloan Kettering Cancer Center, New York, NY 10065, USA;
3. Department of Pediatrics, Memorial Sloan Kettering Cancer Center, New York, NY 10065, USA;
4. Department of Medical Physics, Memorial Sloan Kettering Cancer Center, New York, NY 10065, USA;
5. Department of Pathology, Memorial Sloan Kettering Cancer Center, New York, NY 10065, USA;
6. Tri-Institutional Laboratory of Comparative Pathology, Memorial Sloan Kettering Cancer Center, Weill Cornell Medicine, and The Rockefeller University, New York, NY 10065, USA.
*S.M.L and NK.V.C are Co-PIs on this project.
Abstract
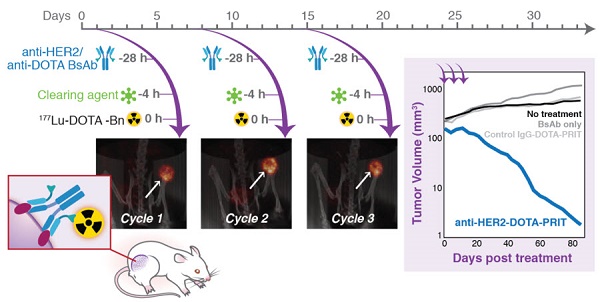
In recent reports, we have shown that optimized pretargeted radioimmunotherapy (PRIT) based on molecularly engineered antibody conjugates and 177Lu-DOTA chelate (DOTA-PRIT) can be used to cure mice bearing human solid tumor xenografts using antitumor antibodies to minimally internalizing membrane antigens, GPA33 (colon) and GD2 (neuroblastoma). However, many solid tumor membrane antigens are internalized after antibody binding and it is generally believed that internalizing tumor membrane antigens are not suitable targets for PRIT. In this study, we tested the hypothesis that DOTA-PRIT can be performed successfully to target HER2, an internalizing membrane antigen widely expressed in breast, ovarian, and gastroesophageal junction cancers.
Methods: DOTA-PRIT was carried out in athymic nude mice bearing BT-474 xenografts, a HER2-expressing human breast cancer, using a three-step dosing regimen consisting of sequential intravenous administrations of: 1) a bispecific IgG-scFv (210 kD) format (BsAb) carrying the IgG sequence of the anti-HER2 antibody trastuzumab and the scFv “C825” with high-affinity, hapten-binding antibody for Bn-DOTA (metal) (BsAb: anti-HER2-C825), 2) a 500 kD dextran-based clearing agent, followed by 3) 177Lu-DOTA-Bn. At the time of treatment, athymic nude mice bearing established subcutaneous BT-474 tumors (medium- and smaller-sized tumors with tumor volumes of 209 ± 101 mm3 and ranging from palpable to 30 mm3, respectively), were studied along with controls. We studied single- and multi-dose regimens. For groups receiving fractionated treatment, we verified quantitative tumor targeting during each treatment cycle using non-invasive imaging with single-photon emission computed tomography/computed tomography (SPECT/CT).
Results: We achieved high therapeutic indices (TI, the ratio of radiation-absorbed dose in tumor to radiation-absorbed dose to critical organs, such as bone marrow) for targeting in blood (TI = 28) and kidney (TI = 7), while delivering average radiation-absorbed doses of 39.9 cGy/MBq to tumor. Based on dosimetry estimates, we implemented a curative fractionated therapeutic regimen for medium-sized tumors that would deliver approximately 70 Gy to tumors, which required treatment with a total of 167 MBq 177Lu-DOTA-Bn/mouse (estimated absorbed tumor dose: 66 Gy). This regimen was well tolerated and achieved 100% complete responses (CRs; defined herein as tumor volume equal to or smaller than 4.2 mm3), including 62.5% histologic cure (5/8) and 37.5% microscopic residual disease (3/8) at 85 days (d). Treatment controls showed tumor progression to 207 ± 201% of pre-treatment volume at 85 d and no CRs. Finally, we show that treatment with this curative 177Lu regimen leads to a very low incidence of histopathologic abnormalities in critical organs such as bone marrow and kidney among survivors compared with non-treated controls.
Conclusion: Contrary to popular belief, we demonstrate that DOTA-PRIT can be successfully adapted to an internalizing antigen-antibody system such as HER2, with sufficient TIs and absorbed tumor doses to achieve a high probability of cures of established human breast cancer xenografts while sparing critical organs of significant radiotoxicity.
Keywords: multistep targeting, bispecific antibodies, HER2, radioimmunotherapy, pretargeting, lutetium-177
 Global reach, higher impact
Global reach, higher impact