13.3
Impact Factor
Theranostics 2018; 8(15):4210-4225. doi:10.7150/thno.25625 This issue Cite
Research Paper
Highly sensitive/selective 3D nanostructured immunoparticle-based interface on a multichannel sensor array for detecting amyloid-beta in Alzheimer's disease
1. Department of Materials Science & Engineering, National Chiao Tung University, No. 1001, Ta-Hsueh Rd., Hsinchu, Taiwan 30010, R.O.C.
2. The Ph.D. Program for Neural Regenerative Medicine, Taipei Medical University, No. 250 Wu-Xing St., Taipei 11010, Taiwan, R.O.C.
3. Department of Biomedical Imaging and Radiological Sciences, National Yang Ming University, No.155, Sec.2, Linong St., Taipei, Taiwan 11221, R.O.C.
4. Department of Nuclear Medicine and National PET/Cyclotron Center, Taipei Veterans General Hospital, No.201, Sec. 2, Shipai Rd., Taipei, Taiwan 11217, R.O.C.
5. Department of Biomedical Engineering, National Yang Ming University, No.155, Sec.2, Linong St., Taipei, Taiwan 11221, R.O.C.
6. Frontier Research Center on Fundamental and Applied Sciences of Matters, National Tsing Hua University, Taiwan 30010, R.O.C.
Abstract
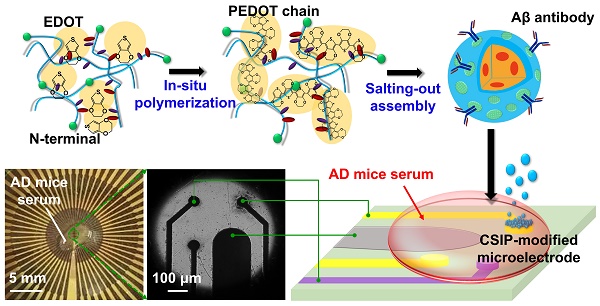
Accumulation of β-amyloid (Aβ) peptides is highly associated with Alzheimer's disease (AD) progression in prevailing studies. The successful development of an ultrasensitive detection assay for Aβ is a challenging task, especially from blood-based samples.
Methods: We have developed a one-step electrophoresis/electropolymerization strategy for preparing a CSIP hierarchical immunoelectrochemical interface that is easily integrated into a PoCT device. The interface includes conductive silk fibroin-based immunoparticles (CSIPs) via electropolymerized Poly(3,4-ethylenedioxythiophene) (PEDOT) bridging to enable on-site electrochemical detection of serum amyloid-β42 (Aβ42) and -β40 (Aβ40) peptides from an AD blood test. In addition, micro-positron emission tomography (microPET) neuroimaging and behavioral tests were simultaneously performed.
Results: This nanostructured conductive interface favors penetration of water-soluble biomolecules and catalyzes a redox reaction, providing limits of detection (LOD) of 6.63 pg/mL for Aβ40 and 3.74 pg/mL for Aβ42. Our proof-of-concept study confirms that the multi-sensing electrochemical immunosensor array (MEIA) platform enables simultaneous measurement of serum Aβ42 and Aβ40 peptide levels and is more informative in early stage AD animals than amyloid-labeling Aβ plaque PET imaging and behavioral tests.
Conclusion: We believe this study greatly expands the applications of silk fibroin-based materials, is an important contribution to the advancement of biomaterials, and would also be valuable in the design of new types of multichannel electrochemical immunosensor arrays for the detection of other diseases.
Keywords: point-of-care testing (PoCT), Alzheimer's disease, amyloid-beta (Aβ), silk fibroin, microPET
 Global reach, higher impact
Global reach, higher impact