13.3
Impact Factor
Theranostics 2018; 8(11):3074-3086. doi:10.7150/thno.24281 This issue Cite
Research Paper
MMP1/PAR1/SP/NK1R paracrine loop modulates early perineural invasion of pancreatic cancer cells
1. Department of Gastroenterology, Sun Yat-sen Memorial Hospital, Sun Yat-sen University, Guangzhou 510120, China
2. Guangdong Provincial Key Laboratory of Malignant Tumor Epigenetics and Gene Regulation, Sun Yat-sen Memorial Hospital, Sun Yat-sen University, Guangzhou 510120, China
3. Department of Gastroenterology, The Seventh Affiliated Hospital of Sun Yat-sen University, Shenzhen 518100, China
4. Department of Gastroenterology, Zhu Hai People's Hospital, JiNan University, Zhuhai 519000, China
5. Department of Radiology, The Fifth Affiliated Hospital of Sun Yat-sen University, Zhuhai 519000, China
6. Department of Hepatobiliary Surgery, The Fifth Affiliated Hospital of Sun Yat-sen University, Zhuhai 519000, China
7. Department of General surgery, The Fifth Affiliated Hospital of Sun Yat-sen University, Zhuhai 519000, China
8. Department of Oncology, The Fifth Affiliated Hospital of Sun Yat-sen University, Zhuhai 519000, China
# These authors contributed equally to this work.
Abstract
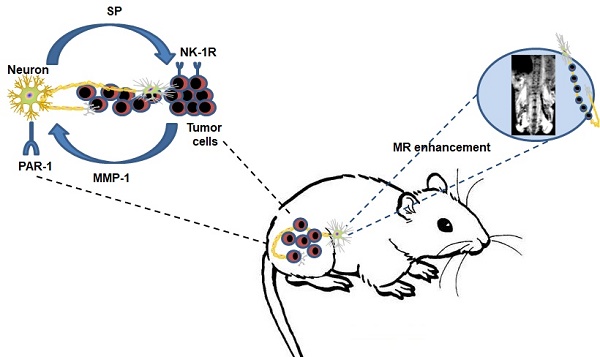
The molecular mechanism of perineural invasion (PNI) is unclear, and insufficient detection during early-stage PNI in vivo hampers its investigation. We aimed to identify a cytokine paracrine loop between pancreatic ductal adenocarcinoma (PDAC) cells and nerves and established a noninvasive method to monitor PNI in vivo.
Methods: A Matrigel/ dorsal root ganglia (DRG) system was used to observe PNI in vitro, and a murine sciatic nerve invasion model was established to examine PNI in vivo. PNI was assessed by MRI with iron oxide nanoparticle labeling. We searched publicly available datasets as well as obtained PDAC tissues from 30 patients to examine MMP1 expression in human tumor and non-tumor tissues.
Results: Our results showed that matrix metalloproteinase-1 (MMP1) activated AKT and induced protease-activated receptor-1 (PAR1)-expressing DRG to release substance P (SP), which, in turn, activated neurokinin 1 receptor (NK1R)-expressing PDAC cells and enhanced cellular migration, invasion, and PNI via SP/NK1R/ERK. In animals, hind limb paralysis and a decreased hind paw width were observed approximately 20 days after inoculation of cancer cells in the perineurium. MMP1 silencing with shRNA or treatment with either a PAR1 or an NK1R antagonist inhibited PNI. MRI detected PNI as early as 10 days after implantation of PDAC cells. PNI also induced PDAC liver metastasis. Bioinformatic analyses and pathological studies on patient tissues corroborated the clinical relevance of these findings.
Conclusion: In this study, we provided evidence that the MMP1/PAR1/SP/NK1R paracrine loop contributes to PNI during the early stage of primary tumor formation. Furthermore, we established a sensitive and non-invasive method to detect nerve invasion using iron oxide nanoparticles and MRI.
Keywords: perineural invasion, iron oxide nanoparticles, MR imaging, metastasis, pancreatic ductal adenocarcinoma
 Global reach, higher impact
Global reach, higher impact