13.3
Impact Factor
Theranostics 2018; 8(11):3007-3021. doi:10.7150/thno.21966 This issue Cite
Research Paper
Flow-dependent epigenetic regulation of IGFBP5 expression by H3K27me3 contributes to endothelial anti-inflammatory effects
1. Aab Cardiovascular Research Institute, Department of Medicine, University of Rochester School of Medicine and Dentistry, Rochester, New York, USA
2. Institute of Medicinal Biotechnology Peking Union Medical College and Chinese Academy of Medical Sciences, Beijing, China
3. Key Laboratory of Fertility Preservation and Maintenance of Ministry of Education, Department of Biochemistry and Molecular Biology, Ningxia Medical University, Yinchuan, China
4. School of Pharmacy, The University of Queensland, Pharmacy Australia Centre of Excellence (PACE), Woolloongabba QLD 4102, Australia
5. Xinhua College of Sun Yat-sen University, Guangzhou, China
6. Department of Vascular and Endovascular Surgery, Klinikum rechts der Isar der Technischen Universitaet Muenchen, Germany
Abstract
Rationale: Atherosclerosis is a chronic inflammatory and epigenetic disease that is influenced by different patterns of blood flow. However, the epigenetic mechanism whereby atheroprotective flow controls endothelial gene programming remains elusive. Here, we investigated the possibility that flow alters endothelial gene expression through epigenetic mechanisms.
Methods: En face staining and western blot were used to detect protein expression. Real-time PCR was used to determine relative gene expression. RNA-sequencing of human umbilical vein endothelial cells treated with siRNA of enhancer of zeste homolog 2 (EZH2) or laminar flow was used for transcriptional profiling.
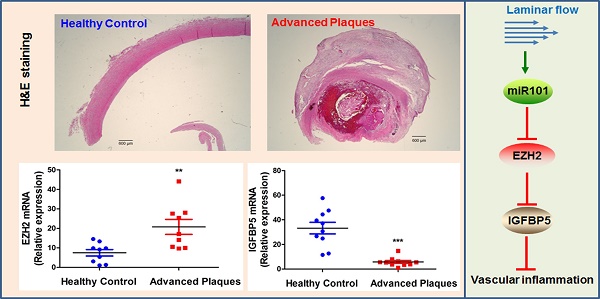
Results: We found that trimethylation of histone 3 lysine 27 (H3K27me3), a repressive epigenetic mark that orchestrates gene repression, was reduced in laminar flow areas of mouse aorta and flow-treated human endothelial cells. The decrease of H3K27me3 paralleled a reduction in the epigenetic “writer”-EZH2, the catalytic subunit of the polycomb repressive complex 2 (PRC2). Moreover, laminar flow decreased expression of EZH2 via mechanosensitive miR101. Genome-wide transcriptome profiling studies in endothelial cells treated with EZH2 siRNA and flow revealed the upregulation of novel mechanosensitive gene IGFBP5 (insulin-like growth factor-binding protein 5), which is epigenetically silenced by H3K27me3. Functionally, inhibition of H3K27me3 by EZH2 siRNA or GSK126 (a specific EZH2 inhibitor) reduced H3K27me3 levels and monocyte adhesion to endothelial cells. Adenoviral overexpression of IGFBP5 also recapitulated the anti-inflammatory effects of H3K27me3 inhibition. More importantly, we observed EZH2 upregulation, and IGFBP5 downregulation, in advanced atherosclerotic plaques from human patients.
Conclusion: Taken together, our findings reveal that atheroprotective flow reduces H3K27me3 as a chromatin-based mechanism to augment the expression of genes that confer an anti-inflammatory response in the endothelium. Our study exemplifies flow-dependent epigenetic regulation of endothelial gene expression, and also suggests that targeting the EZH2/H3K27me3/IGFBP5 pathway may offer novel therapeutics for inflammatory disorders such as atherosclerosis.
Keywords: atherosclerosis, endothelial cells, epigenetic, EZH2, H3K27me3, IGFBP5
 Global reach, higher impact
Global reach, higher impact