Impact Factor
Theranostics 2018; 8(10):2765-2781. doi:10.7150/thno.22904 This issue Cite
Research Paper
Placenta-specific drug delivery by trophoblast-targeted nanoparticles in mice
1. Laboratory for Reproductive Health, Institute of Biomedicine and Biotechnology, Shenzhen Institutes of Advanced Technology (SIAT), Chinese Academy of Sciences (CAS), Shenzhen, Guangdong, China, 518055.
2. College of Veterinary Medicine, Hunan Agricultural University, Changsha, Hunan, China, 410128.
3. Bao'an Maternal and Child Health Care Hospital, Shenzhen, China, 518133.
4. John A. Moran Eye Center, University of Utah, Salt Lake City, Utah, USA, 84132.
5. Guangdong Key Laboratory of Nanomedicine, CAS Key Lab for Health Informatics, Institute of Biomedicine and Biotechnology, Shenzhen Institutes of Advanced Technology, Chinese Academy of Sciences, Shenzhen, China, 518055.
6. Department of Obstetrics and Gynecology, Wayne State University School of Medicine, C.S. Mott Center for Human Growth and Development, Detroit, Michigan, USA, 48201.
* Contributed equally to this work
Abstract
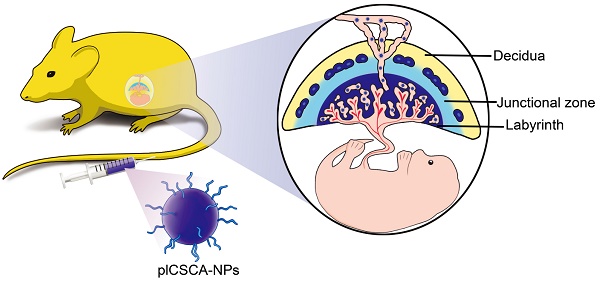
Rationale: The availability of therapeutics to treat pregnancy complications is severely lacking, mainly due to the risk of harm to the fetus. In placental malaria, Plasmodium falciparum-infected erythrocytes (IEs) accumulate in the placenta by adhering to chondroitin sulfate A (CSA) on the surfaces of trophoblasts. Based on this principle, we have developed a method for targeted delivery of payloads to the placenta using a synthetic placental CSA-binding peptide (plCSA-BP) derived from VAR2CSA, a CSA-binding protein expressed on IEs.
Methods: A biotinylated plCSA-BP was used to examine the specificity of plCSA-BP binding to mouse and human placental tissue in tissue sections in vitro. Different nanoparticles, including plCSA-BP-conjugated nanoparticles loaded with indocyanine green (plCSA-INPs) or methotrexate (plCSA-MNPs), were administered intravenously to pregnant mice to test their efficiency at drug delivery to the placenta in vivo. The tissue distribution and localization of the plCSA-INPs were monitored in live animals using an IVIS imaging system. The effect of plCSA-MNPs on fetal and placental development and pregnancy outcome were examined using a small-animal high-frequency ultrasound (HFUS) imaging system, and the concentrations of methotrexate in fetal and placental tissues were measured using high-performance liquid chromatography (HPLC).
Results: plCSA-BP binds specifically to trophoblasts and not to other cell types in the placenta or to CSA-expressing cells in other tissues. Moreover, we found that intravenously administered plCSA-INPs accumulate in the mouse placenta, and ex vivo analysis of the fetuses and placentas confirmed placenta-specific delivery of these nanoparticles. We also demonstrate successful delivery of methotrexate specifically to placental cells by plCSA-BP-conjugated nanoparticles, resulting in dramatic impairment of placental and fetal development. Importantly, plCSA-MNPs treatment had no apparent adverse effects on maternal tissues.
Conclusion: These results demonstrate that plCSA-BP-guided nanoparticles could be used for the targeted delivery of payloads to the placenta and serve as a novel placenta-specific drug delivery option.
Keywords: trophoblast, chondroitin sulfate A, placental CSA binding peptide, nanoparticles
 Global reach, higher impact
Global reach, higher impact