13.3
Impact Factor
Theranostics 2018; 8(2):549-562. doi:10.7150/thno.20047 This issue Cite
Research Paper
A cell-penetrating whole molecule antibody targeting intracellular HBx suppresses hepatitis B virus via TRIM21-dependent pathway
1. State Key Laboratory of Molecular Vaccinology and Molecular Diagnostics, School of Public Health, Xiamen University, Xiamen 361102, China;
2. National Institute of Diagnostics and Vaccine Development in Infectious Diseases, School of Life Science, Xiamen University, Xiamen 361102, China;
3. School of Medicine, Shenzhen University, Shenzhen, 518060, China;
4. Department of Clinical Pathology, Affiliated Hospital of Guangdong Medical College, Zhanjiang, 524001, China.
* These authors contributed equally to this work
Abstract
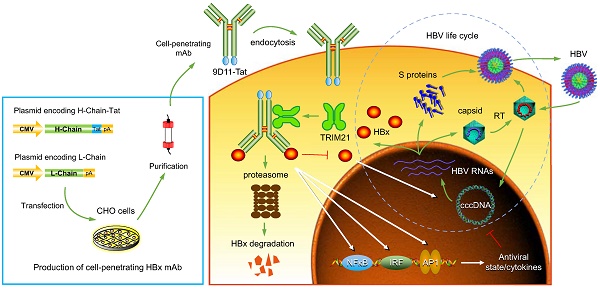
Rationale: Monoclonal antibodies (mAbs) mostly targeting extracellular or cell surface molecules have been widely used in the treatment of various diseases. However, mAbs cannot pass through the cell membrane as efficiently as small compounds, thus limiting their use against intracellular targets. Methods to shuttle antibodies into living cells may largely expand research and application in areas based on mAbs. Hepatitis B virus X protein (HBx) is an important intracellular multi-functional viral protein in the life cycle of hepatitis B virus (HBV). HBx plays essential roles in virus infection and replication and is strongly associated with HBV-related carcinogenesis.
Methods: In this study, we developed a cell-penetrating whole molecule antibody targeting HBx (9D11-Tat) by the fusion of a cell penetrating peptide (CPP) on the C-terminus of the heavy chain of a potent mAb specific to HBx (9D11). The anti-HBV effect and mechanism of 9D11-Tat were investigated in cell and mouse models mimicking chronic HBV infection.
Results: Our results demonstrated that the recombinant 9D11-Tat antibody could efficiently internalize into living cells and significantly suppress viral transcription, replication, and protein production both in vitro and in vivo. Further analyses suggested the internalized 9D11-Tat antibody could greatly reduce intracellular HBx via Fc binding receptor TRIM21-mediated protein degradation. This process simultaneously stimulated the activations of NF-κB, AP-1, and IFN-β, which promoted an antiviral state of the host cell.
Conclusion: In summary, our study offers a new approach to target intracellular pathogenesis-related protein by engineered cell-penetrating mAb expanding their potential for therapeutic applications. Moreover, the 9D11-Tat antibody may provide a novel therapeutic agent against human chronic HBV infection.
Keywords: antibodies for intracellular targets, cell-penetrating peptide, antibody-mediated intracellular immunity, hepatitis B virus, Hepatitis B virus X protein
 Global reach, higher impact
Global reach, higher impact