Impact Factor
Theranostics 2018; 8(2):423-436. doi:10.7150/thno.22377 This issue Cite
Research Paper
Methylation-associated silencing of miR-193a-3p promotes ovarian cancer aggressiveness by targeting GRB7 and MAPK/ERK pathways
1. Department of Obstetrics & Gynaecology, LKS Faculty of Medicine, The University of Hong Kong, Hong Kong SAR, People's Republic of China;
2. Faculty of Medical Laboratory Science, Rujin Hospital, School of Medicine, Shanghai Jiao Tong University, Shanghai 200030, People's Republic of China;
3. Department of Gynecological Oncology, Fudan University Shanghai Cancer Center, Fudan University, Shanghai, 200032, People's Republic of China;
4. Department of Oncology, Shanghai Medical College, Fudan University, Shanghai, 200032, People's Republic of China.
Abstract
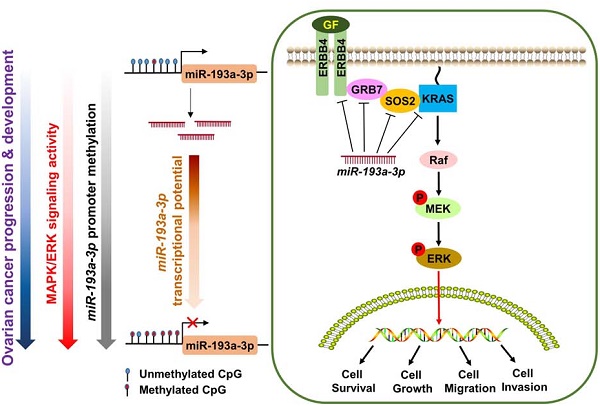
Human growth factor receptor-bound protein-7 (GRB7) is a pivotal mediator involved in receptor tyrosine kinase signaling and governing diverse cellular processes. Aberrant upregulation of GRB7 is frequently associated with the progression of human cancers. However, the molecular mechanisms leading to the upregulation of GRB7 remain largely unknown. Here, we propose that the epigenetic modification of GRB7 at the post-transcriptional level may be a crucial factor leading to GRB7 upregulation in ovarian cancers. Methods: The upstream miRNA regulators were predicted by in silico analysis. Expression of GRB7 was examined by qPCR, immunoblotting and immunohistochemical analyses, while miR-193a-3p levels were evaluated by qPCR and in situ hybridization in ovarian cancer cell lines and clinical tissue arrays. MS-PCR and pyrosequencing analyses were used to assess the methylation status of miR-193a-3p. Stable overexpression or gene knockdown and Tet-on inducible approaches, in combination with in vitro and in vivo tumorigenic assays, were employed to investigate the functions of GRB7 and miR-193a-3p in ovarian cancer cells. Results: Both miR-193a-3p and its isoform, miR-193b-3p, directly targeted the 3' UTR of GRB7. However, only miR-193a-3p showed a significantly inverse correlation with GRB7-upregulated ovarian cancers. Epigenetic studies revealed that methylation-mediated silencing of miR-193a-3p led to a stepwise decrease in miR-193a-3p expression from low to high-grade ovarian cancers. Intriguingly, miR-193a-3p not only modulated GRB7 but also ERBB4, SOS2 and KRAS in the MAPK/ERK signaling pathway to enhance the oncogenic properties of ovarian cancer cells in vitro and in vivo. Conclusion: These findings suggest that epigenetic silencing of miR-193a-3p by DNA hypermethylation is a dynamic process in ovarian cancer progression, and miR-193a-3p may be explored as a promising miRNA replacement therapy in this disease.
Keywords: DNA hypermethylation, miR-193a-3p, GRB7, MAPK signaling, ovarian cancer.
 Global reach, higher impact
Global reach, higher impact