13.3
Impact Factor
Theranostics 2017; 7(19):4699-4709. doi:10.7150/thno.20654 This issue Cite
Research Paper
Glycan and Peptide IgE Epitopes of the TNF-alpha Blockers Infliximab and Adalimumab - Precision Diagnostics by Cross-Reactivity Immune Profiling of Patient Sera
1. Clinical and Molecular Allergology, Priority Research Area Asthma & Allergy, Airway Research Center North (ARCN), German Center for Lung Research (DZL), Research Center Borstel, Borstel, Germany;
2. Mucosal Immunology and Diagnostics, Priority Research Area Asthma & Allergy, Airway Research Center North (ARCN), German Center for Lung Research (DZL), Research Center Borstel, Borstel, Germany;
3. IPM Biotech, Hamburg, Germany;
4. Department of Medicine, Division of Allergy and Clinical Immunology, University of Virginia School of Medicine, Charlottesville, USA;
5. Interdisciplinary Allergy Outpatient Clinic, Department of Internal Medicine, University of Lübeck, Lübeck, Germany.
Abstract
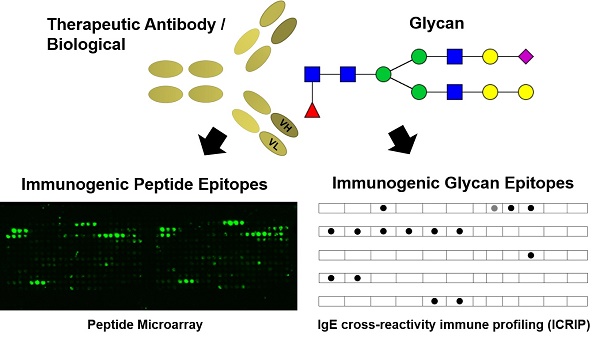
Biological drugs like therapeutic antibodies are widely used for the treatment of various diseases like inflammatory disorders and cancer. A drawback of these novel treatments is the substantial proportion of patients experiencing adverse reactions such as loss-of-drug effect or hypersensitivity reactions. These reactions are associated with pre-existing and/or developing anti-drug antibodies. Especially IgE development is a risk factor for life-threatening systemic anaphylaxis.
Methods: In order to characterize the individual drug-specific serum IgE, an IgE cross-reactivity immune profiling (ICRIP) assay was developed. Individual IgG epitopes of anti-drug antibodies against adalimumab were identified by epitope mapping via peptide microarray.
Results: ICRIP analyses of sera from patients treated with the therapeutic antibodies adalimumab (ADL) and infliximab (IFX) reveal individual, distinct IgE binding patterns. IgG epitopes were identified mostly located in the variable region of ADL.
Conclusions: Using ICRIP and peptide microarrays for pharmacovigilance of the TNF-α blockers IFX and ADL, risk factors and biomarkers before and during therapy shall be identified. These diagnostic systems provide the basis for a safe and efficacious therapy decision for each patient in cases of adverse drug reactions mediated by different types of anti-drug antibodies.
Keywords: adalimumab, anti-drug antibodies, hypersensitivity, infliximab, precision medicine.
 Global reach, higher impact
Global reach, higher impact