13.3
Impact Factor
Theranostics 2017; 7(15):3715-3731. doi:10.7150/thno.19678 This issue Cite
Research Paper
Selective Targeting of a Novel Vasodilator to the Uterine Vasculature to Treat Impaired Uteroplacental Perfusion in Pregnancy
1. Maternal and Fetal Health Research Centre, Division of Developmental Biology and Medicine, University of Manchester, Manchester, UK;
2. Academic Health Science Centre, St Mary's Hospital, Oxford Road, Manchester, M13 9WL, UK;
3. Cancer Center, Sanford-Burnham Medical Research Institute, 10901 N. Torrey Pines Road, La Jolla, CA 92037, USA and Center for Nanomedicine and Department of Cell, Molecular and Developmental Biology, University of California, Santa Barbara, Santa Barbara, CA 93106-9610, USA;
4. Laboratory of Cancer Biology, Institute of Biomedicine, Centre of Excellence for Translational Medicine, University of Tartu, Tartu, Estonia;
5. Dipartimento di Chimica, Materiali ed Ingegneria Chimica "G. Natta". Politecnico di Milano, Via Mancinelli 7, 20131 Milan, Italy;
6. Fondazione CEN - European Centre for Nanomedicine, Piazza Leonardo da Vinci 32, 20133 Milan, Italy;
7. Division of Pharmacy and Optometry, University of Manchester, Stopford Building, Oxford Road, Manchester, M13 9PT, UK.
Abstract
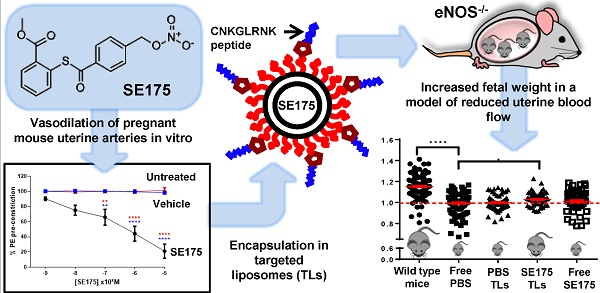
Fetal growth restriction (FGR) in pregnancy is commonly caused by impaired uteroplacental blood flow. Vasodilators enhance uteroplacental perfusion and fetal growth in humans and animal models; however, detrimental maternal and fetal side effects have been reported. We hypothesised that targeted uteroplacental delivery of a vasodilator would enhance drug efficacy and reduce the risks associated with drug administration in pregnancy. Phage screening identified novel peptides that selectively accumulated in the uteroplacental vasculature of pregnant mice. Following intravenous injection, the synthetic peptide CNKGLRNK selectively bound to the endothelium of the uterine spiral arteries and placental labyrinth in vivo; CNKGLRNK-decorated liposomes also selectively bound to these regions. The nitric oxide donor 2-[[4-[(nitrooxy)methyl]benzoyl]thio]-benzoic acid methyl ester (SE175) induced significant relaxation of mouse uterine arteries and human placental arteries in vitro; thus, SE175 was encapsulated into these targeted liposomes and administered to healthy pregnant C57BL/6J mice or endothelial nitric oxide synthase knockout (eNOS-/-) mice, which exhibit impaired uteroplacental blood flow and FGR. Liposomes containing SE175 (0.44mg/kg) or PBS were administered on embryonic (E) days 11.5, 13.5, 15.5 and 17.5; fetal and placental weights were recorded at term and compared to mice injected with free PBS or SE175. Targeted uteroplacental delivery of SE175 had no effect on fetal weight in C57BL/6J mice, but significantly increased fetal weight and mean spiral artery diameter, and decreased placental weight, indicative of improved placental efficiency, in eNOS-/- mice; free SE175 had no effect on fetal weight or spiral artery diameter. Targeted, but not free SE175 also significantly reduced placental expression of 4-hydroxynonenal, cyclooxygenase-1 and cyclooxygenase-2, indicating a reduction in placental oxidative stress. These data suggest that exploiting vascular targeting peptides to selectively deliver SE175 to the uteroplacental vasculature may represent a novel treatment for FGR resulting from impaired uteroplacental perfusion.
Keywords: pregnancy, placenta, nitric oxide, endothelium, vascular biology, drug targeting.
 Global reach, higher impact
Global reach, higher impact