Impact Factor
Theranostics 2017; 7(10):2718-2731. doi:10.7150/thno.19662 This issue Cite
Research Paper
Focused Ultrasound Hyperthermia Mediated Drug Delivery Using Thermosensitive Liposomes and Visualized With in vivo Two-Photon Microscopy
1. Physical Sciences Platform, Sunnybrook Research Institute, Toronto, ON, Canada;
2. Department of Medical Biophysics, University of Toronto, Toronto, ON, Canada;
3. Institute of Biomaterials and Biomedical Engineering, University of Toronto, Toronto, ON, Canada.
Abstract
The future of nanomedicines in oncology requires leveraging more than just the passive drug accumulation in tumors through the enhanced permeability and retention effect. Promising results combining mild hyperthermia (HT) with lyso-thermosensitive liposomal doxorubicin (LTSL-DOX) has led to improved drug delivery and potent antitumor effects in pre-clinical studies. The ultimate patient benefit from these treatments can only be realized when robust methods of HT can be achieved clinically. One of the most promising methods of non-invasive HT is the use of focused ultrasound (FUS) with MRI thermometry for anatomical targeting and feedback. MRI-guided focused ultrasound (MRgFUS) is limited by respiratory motion and large blood vessel cooling. In order to translate exciting pre-clinical results to the clinic, novel heating approaches capable of overcoming the limitations on clinical MRgFUS+HT must be tested and evaluated on their ability to locally release drug from LTSL-DOX.
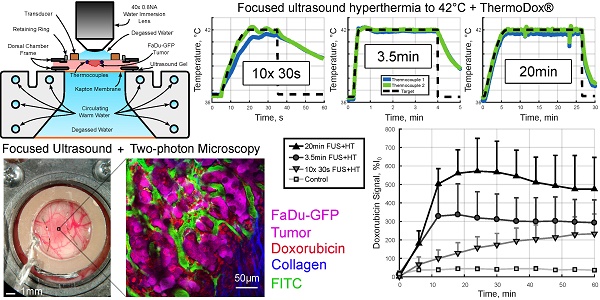
Methods: In this work, a new system is described to integrate focused ultrasound (FUS) into a two-photon microscopy (2PM) setting to image the release of drug from LTSL-DOX in real-time during FUS+HT in vivo. A candidate scheme for overcoming the limitations of respiratory motion and large blood vessel cooling during MRgFUS+HT involves applying FUS+HT to 42°C in short ~30s bursts. The spatiotemporal drug release pattern from LTSL-DOX as a result is quantified using 2PM and compared against continuous (3.5min and 20min at 42°C) FUS+HT schemes and unheated controls.
Results: It was observed for the first time in vivo that these short duration temperature elevations could produce substantial drug release from LTSL-DOX. Ten 30s bursts of FUS+HT was able to achieve almost half of the interstitial drug concentration as 20min of continuous FUS+HT. There was no significant difference between the intravascular area under the concentration-time curve for ten 30s bursts of FUS+HT and 3.5min of continuous FUS+HT.
Conclusion: We have successfully combined 2PM with FUS+HT for imaging the release of DOX from LTSL-DOX in vivo in real-time, which will permit the investigation of FUS+HT heating schemes to improve drug delivery from LTSL-DOX. We have evaluated the ability to release DOX in short 30s FUS+HT bursts to 42°C as a method to overcome limitations on clinical MRgFUS+HT and have found that such exposures are capable of releasing measurable amounts of drug. Such an exposure has the potential to overcome limitations that hamper conventional MRgFUS+HT treatments in targets that are associated with substantial tissue motion.
Keywords: Focused ultrasound hyperthermia, two-photon microscopy, thermosensitive liposomes, heat-targeted cancer therapy, doxorubicin.
 Global reach, higher impact
Global reach, higher impact