13.3
Impact Factor
Theranostics 2017; 7(8):2164-2176. doi:10.7150/thno.19652 This issue Cite
Research Paper
Vandetanib-eluting Radiopaque Beads: In vivo Pharmacokinetics, Safety and Toxicity Evaluation following Swine Liver Embolization
1. Department of Radiology and Interventional Radiology, CHUV University of Lausanne, Lausanne, Switzerland;
2. Biocompatibles UK Ltd, a BTG International group company, Lakeview, Riverside Way, Watchmoor Park, Camberley, GU15 3YL, UK.
Abstract
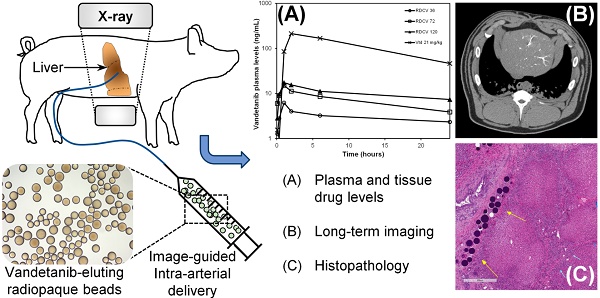
PURPOSE: To evaluate the plasma and tissue pharmacokinetics, safety and toxicity following intra-arterial hepatic artery administration of Vandetanib (VTB)-eluting Radiopaque Beads (VERB) in healthy swine.
MATERIALS AND METHODS: In a first phase, healthy swine were treated with hepatic intra-arterial administration of VERB at target dose loading strengths of 36 mg/mL (VERB36), 72 mg/mL (VERB72) and 120 mg/mL (VERB120). Blood and tissue samples were taken and analysed for VTB and metabolites to determine pharmacokinetic parameters for the different dose forms over 30 days. In a second phase, animals were treated with unloaded radiopaque beads or high dose VTB loaded beads (VERB100, 100 mg/mL). Tissue samples from embolized and non-embolized areas of the liver were evaluated at necropsy (30 and 90 days) for determination of VTB and metabolite levels and tissue pathology. Imaging was performed prior to sacrifice using multi-detector computed tomography (MDCT) and imaging findings correlated with pathological changes in the tissue and location of the radiopaque beads.
RESULTS: The peak plasma levels of VTB (Cmax) released from the various doses of VERB ranged between 6.19-17.3 ng/mL indicating a low systemic burst release. The plasma profile of VTB was consistent with a distribution phase up to 6 h after administration followed by elimination with a half-life of 20-23 h. The AUC of VTB and its major metabolite N-desmethyl vandetanib (NDM VTB) was approximately linear with the dose strength of VERB. VTB plasma levels were at or below limits of detection two weeks after administration. In liver samples, VTB and NDM VTB were present in treated sections at 30 days after administration at levels above the in vitro IC50 for biological effectiveness. At 90 days both analytes were still present in treated liver but were near or below the limit of quantification in untreated liver sections, demonstrating sustained release from the VERB. Comparison of the reduction of the liver lobe size and associated tissue changes suggested a more effective embolization with VERB compared to the beads without drug.
CONCLUSIONS: Hepatic intra-arterial administration of VERB results in a low systemic exposure and enables sustained delivery of VTB to target tissues following embolization. Changes in the liver tissue are consistent with an effective embolization and this study has demonstrated that VERB100 is well tolerated with no obvious systemic toxicity.
Keywords: Vandetanib-eluting Radiopaque Beads, pharmacokinetics, safety and toxicity
 Global reach, higher impact
Global reach, higher impact