13.3
Impact Factor
Theranostics 2017; 7(5):1290-1302. doi:10.7150/thno.18557 This issue Cite
Research Paper
Design of Tumor Acidity-Responsive Sheddable Nanoparticles for Fluorescence/Magnetic Resonance Imaging-Guided Photodynamic Therapy
1. School of Biological and Medical Engineering, Hefei University of Technology, Hefei, Anhui 230009, China;
2. Division of Gastroenterology, Affiliated Provincial Hospital, Anhui Medical University, Anhui 230001, China;
3. Department of Oncolog, the First Affiliated Hospital of Anhui Medical University, Hefei 230022, China;
4. The First Hospital and Institute of Immunology, Jilin University, Changchun 130061, P. R. China;
5. Department of Radiology, The First Hospital of JiLin University, Changchun, 130061, P. R. China.
* These authors contributed equally to this work.
Abstract
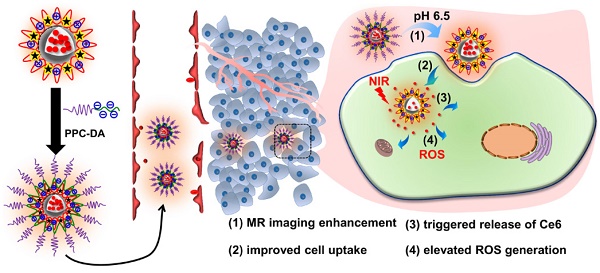
Imaging-guided cancer therapy, which integrates diagnostic and therapeutic functionalities into a single system, holds great promise to enhance the accuracy of diagnosis and improve the efficacy of therapy. Specifically, for photodynamic therapy (PDT), it is highly desirable to precisely focus laser light onto the tumor areas to generate reactive oxygen species (ROS) that are cytotoxic tumor cells and avoid light-associated side effects. Herein, a distinct three-layer nanostructured particle with tumor acidity-responsiveness (S-NP) that encapsulates the photosensitizer chlorin e6 (Ce6) and chelates Gd3+ is successfully developed for fluorescence/magnetic resonance (MR) dual-model imaging-guided precision PDT. We show clear evidence that the outer PEG layer significantly prolongs circulation time, and the inner poly(ε-caprolactone) (PCL) core can physically encapsulate Ce6. More interestingly, the middle layer of the S-NP, acting as a molecular fence to keep Ce6 in the circulation system, was dismantled by the slightly acidic tumor microenvironment. Afterwards, the PEG shell is deshielded from the S-NP at the tumor tissue, resulting in improved cell uptake, enlarged MR signal intensity, rapid release of Ce6 within tumor cells, and elevated PDT efficacy. Our results suggest that tumor-acidity-responsive nanoparticles with fine design could serve as a theranostic platform with great potential in imaging-guided PDT treatment of cancer.
Keywords: sheddable nanoparticles, tumor acidity-responsive, photodynamic therapy, magnetic resonance imaging
 Global reach, higher impact
Global reach, higher impact