13.3
Impact Factor
Theranostics 2017; 7(4):926-934. doi:10.7150/thno.17131 This issue Cite
Research Paper
Visualization of Macrophage Recruitment to Inflammation Lesions using Highly Sensitive and Stable Radionuclide-Embedded Gold Nanoparticles as a Nuclear Bio-Imaging Platform
1. Department of Nuclear Medicine, Kyungpook National University Hospital, Daegu, South Korea;
2. Leading-edge Research Center for Drug Discovery and Development for Diabetes and Metabolic Disease, Kyungpook National University Hospital, Daegu, South Korea;
3. Department of Pathology, Chemon Co. Ltd., 240, Nampyeong-Ro, Yangji-Myeon, Cheoin-Gu, Yongin-Si, Gyeonggi-Do, 17162, Republic of Korea;
4. New Drug Development Center, Daegu-Gyeongbuk Medical Innovation Foundation, Daegu, South Korea;
5. Department of Pharmacy, School of Pharmacy, Massachusetts College of Pharmacy and Health Sciences, Boston, Massachusetts, USA;
6. Department of Internal Medicine, Kyungpook National University School of Medicine, Deagu 700-721, South Korea;
7. KU-KIST Graduate School of Converging Science and Technology, Korea University, Seoul Anam-ro 145, South Korea;
8. Daegu-Gyeongbuk Medical Innovation Foundation, Daegu, South Korea;
9. Laboratory Animal Center, Daegu-Gyeongbuk Medical Innovation Foundation, Daegu, South Korea.
*The first two authors contributed equally to this study.
Abstract
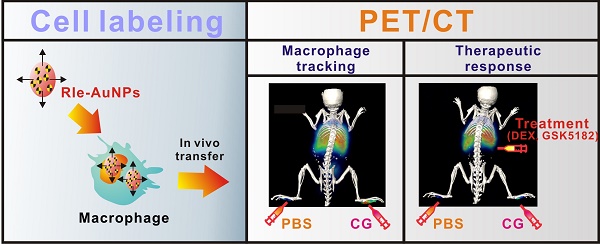
Reliable and sensitive imaging tools are required to track macrophage migration and provide a better understating of their biological roles in various diseases. Here, we demonstrate the possibility of radioactive iodide-embedded gold nanoparticles (RIe-AuNPs) as a cell tracker for nuclear medicine imaging. To demonstrate this utility, we monitored macrophage migration to carrageenan-induced sites of acute inflammation in living subjects and visualized the effects of anti-inflammatory agents on this process. Macrophage labeling with RIe-AuNPs did not alter their biological functions such as cell proliferation, phenotype marker expression, or phagocytic activity. In vivo imaging with positron-emission tomography revealed the migration of labeled macrophages to carrageenan-induced inflammation lesions 3 h after transfer, with highest recruitment at 6 h and a slight decline of radioactive signal at 24 h; these findings were highly consistent with the data of a bio-distribution study. Treatment with dexamethasone (an anti-inflammation drug) or GSK5182 (an ERRγ inverse agonist) hindered macrophage recruitment to the inflamed sites. Our findings suggest that a cell tracking strategy utilizing RIe-AuNPs will likely be highly useful in research related to macrophage-related disease and cell-based therapies.
Keywords: gold nanoparticles, nuclear bio-imaging platform, macrophage migration, acute inflammation.
 Global reach, higher impact
Global reach, higher impact