Impact Factor
Theranostics 2017; 7(4):826-845. doi:10.7150/thno.17071 This issue Cite
Review
Progress of small molecular inhibitors in the development of anti-influenza virus agents
1. Department of Nuclear Medicine, West China Hospital, Sichuan University, Chengdu, Sichuan Province, China.
2. State Key Laboratory of Biotherapy and Cancer Center, West China Hospital, and Collaborative Innovation Center for Biotherapy, Sichuan University, Sichuan 610041, China.
3. Department of Laboratory Medicine, West China Second University Hospital, Sichuan University.
4. Key Laboratory of Birth Defects and Related Diseases of Women and Children (Sichuan University), Ministry of Education.
* These authors contributed equally to this work.
Received 2016-8-1; Accepted 2016-11-18; Published 2017-2-8
Abstract
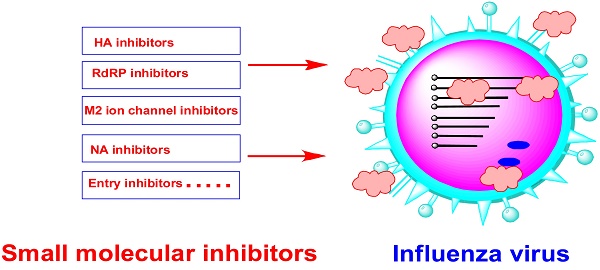
The influenza pandemic is a major threat to human health, and highly aggressive strains such as H1N1, H5N1 and H7N9 have emphasized the need for therapeutic strategies to combat these pathogens. Influenza anti-viral agents, especially active small molecular inhibitors play important roles in controlling pandemics while vaccines are developed. Currently, only a few drugs, which function as influenza neuraminidase (NA) inhibitors and M2 ion channel protein inhibitors, are approved in clinical. However, the acquired resistance against current anti-influenza drugs and the emerging mutations of influenza virus itself remain the major challenging unmet medical needs for influenza treatment. It is highly desirable to identify novel anti-influenza agents. This paper reviews the progress of small molecular inhibitors act as antiviral agents, which include hemagglutinin (HA) inhibitors, RNA-dependent RNA polymerase (RdRp) inhibitors, NA inhibitors and M2 ion channel protein inhibitors etc. Moreover, we also summarize new, recently reported potential targets and discuss strategies for the development of new anti-influenza virus drugs.
Keywords: anti-influenza virus agent, hemagglutinin inhibitors, RNA-dependent RNA polymerase inhibitors, neuraminidase inhibitors, M2 ion channel protein inhibitors.
Introduction
The influenza virus continues to threaten public health because of its high morbidity and mortality rates despite the efforts and success of antiviral research (1-3). According to the World Health Organization (WHO), seasonal influenza virus epidemics result in the infection of 3-5 million people and 250,000 to 500,000 deaths worldwide (4). In recent years, the emergence of highly aggressive virus strains such as H1N1, H5N1, and H7N9 has reemphasized the need for therapeutic strategies to overcome these pathogens (5). Vaccines and antiviral agents are essential for mitigating the influence of the influenza virus (6, 7). Due to the frequent variation in the influenza virus, anti-influenza agents seem to be more effective at preventing the highly contagious infection with the virus and at treating disease epidemics.
Influenza pandemics are caused by the influenza virus, a negative-sense single-stranded member of the Orthomyxoviridae family of RNA viruses (8). Influenza virus could be classified as one of three distinct subtypes (influenza A, influenza B, or influenza C) depending on its nucleoproteins and the antigen determinants of its matrix proteins. Influenza A and influenza B appear to cause highly infectious diseases, while influenza C does not seem to cause significant disease (1). However, pandemic outbreaks are caused by influenza A viruses. Therefore, much more attention has been paid to the influenza A viruses.
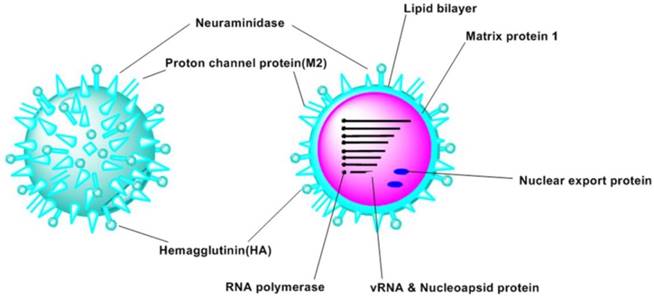
The structure of the influenza virus contains three motifs (Figure 1): the core, the matrix protein and the viral envelope (9). Influenza A virus was made up by proteins encoded by eight segments of negative-strand RNA (8, 10). These proteins include hemagglutinin (HA), neuraminidase (NA), nucleoprotein (NP), RNA polymerase (PA, PB1, PB2), matrix protein 1 (M1), proton channel protein (M2), non-structural protein 1 (NS1) and nuclear export protein (NEP, NS2) (8, 11). Additionally, some proteins exist in particular strains were identified recently, such as PB1-F2 (12), PB1-N40 (10), and PA-X (13). Moreover, some novel proteins have been identified recently (14).
Depending on the differences of the subtypes of 18 HA (H1-H18) and 11 NA (N1-N11), influenza A viruses can be classified into 162 subtypes (15).
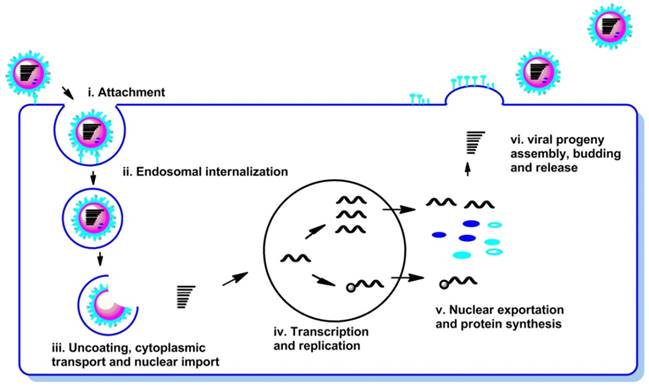
The life cycle of the influenza virus is a complex biological process and can be divided into the following steps (Figure 2): (i) attachment of the virion to the cell surface (receptor binding); (ii) endosomal internalization of the virus into cell (endocytosis and endocytosis); (iii) uncoating, cytoplasmic transport and nuclear import of viral ribonucleoproteins (vRNPs); (iv) transcription and replication of the viral RNAs; (v) nuclear exportation and protein synthesis; (vi) viral progeny assembly, budding and release from the cell membrane.
Viral proteins and host cellular proteins involved in each step of the life cycle of virus infection are attractive target to combat influenza virus infections (8, 16, 17).
Schematic views of the structure of influenza A virus.
Schematic representation of the life cycle of influenza virus.
Structures of currently available licensed anti-influenza drugs.
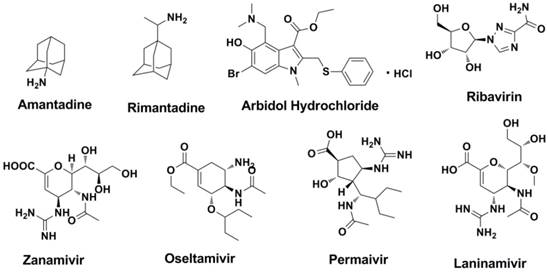
At present, only a few drugs that function as influenza NA inhibitors, M2 ion channel protein inhibitors, RNA-dependent RNA polymerase inhibitors and protease inhibitors are used in clinical. Structures of currently available licensed anti-influenza drugs were shown in Figure 3.
The emergence of influenza virus mutations and acquired resistance are extremely influential in driving the development of new anti-influenza agents against possible future influenza epidemics. Small molecular inhibitors (molecular weight less than 1000) are powerful tools to fight against influenza virus. Besides small molecular inhibitors, peptides (Entry Block-peptide (EB-peptide) (18, 19), FluPep (FP) peptides (20) , NDFRSKT (21, 22) etc.), proteins (Hepatocyte growth factor activator inhibitor 2 (23), Ulinastatin (23-25) and Fludase (26-28)), monoclonal antibodies (29) , Nanoparticles (30-33) and other types of anti-viral drugs are effective in influenza virus infection (those agents are out of the scope of this article). According to the target of the antiviral agent, research on anti-influenza virus agents can currently be categorized into two fields: agents that target functional proteins of the virus itself and agents that target potential sites of the host cells.
1. Agents targeting functional proteins of the virus itself
The HA protein facilitates viral binding to receptors on host cells receptors and the fusion process. Viral RNA replication and transcription is carried out by the nucleoprotein (NP) and three polymerase subunits (PB2, PB1 and PA). The M2 protein is involved in uncoating and maturation of the virus. The NA protein is essential for the release of the virus from infected cells. Therefore, the functional proteins described above are attractive targets for antiviral research.
1.1 Entry inhibitors
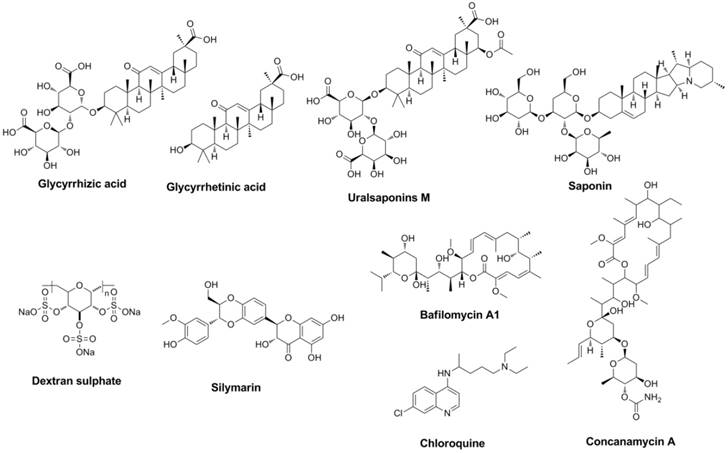
Antiviral compounds designed (or discovered) to interrupt the attachment and entry course of the virus were named entry inhibitors (34). Extracted from nature produce from Traditional Chinese Medicine (TCM) licorice, triterpenoids derivatives showed antiviral activities (35-40). Glycyrrhizic acid (or named glycyrrhizin) (41) and glycyrrhetinic acid (42) have been proved to be effective to interrupt the attachment and entry course of the influenza virus (Figure 4).
Glycyrrhizin exhibited broad spectrum inhibitory activity and showed the most potent inhibitory activity against the replication of H3N2, H5N1 etc (43). Saponins and uralsaponins M-Y (Figure 4) showed anti-influenza activities in recent studies (44). Uralsaponins M exhibited inhibitory activities against influenza virus A/WSN/33 H1N1 in MDCK cell lines with IC50 value of 48.0 μM (44).
As a negatively charged sulphated polysaccharide, Dextran sulphate (DS, Figure 4) inhibit the attachment and entry course as well as the HA-dependent (45, 46) and NA-dependent (47) fusion course (strictly speaking, DS is not a small molecular). Silymarin (Figure 4) (48), extract from milk thistle seeds, showed activity against influenza. Its main component silibinin and derivatives can also regulate autophagy course. As a multi-target natural product, curcumin can also inhibit the virus entry course and HA activity (EC50 was approximately 0.47 µM for inhibition of influenza virus, Plaque Reduction Assay) (49-51). Lysosomotropic agents (such as Concanamycin A, Bafilomycin A1, Chloroquine etc.), exhibit their anti-influenza activities depending on the pH value of the intracellular environment (52-57).
Structures of representative entry inhibitors.
1.2 Hemagglutinin (HA) inhibitors
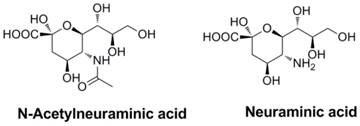
HA is encoded by the vRNAs and is composed of three identical structural subunits (1). These subunits have two important functions in linkage and the internalization trigger: first, HA can provide a link between the virus and the surface of target host cells (58-60); and second, HA triggers the internalization of the virus through the fusion of the viral envelope and the endosomal membrane of the host. Both HA and NA recognize N-acetylneuraminic acid (Neu5Ac, also called sialic acid), a typical terminal unit of glycoconjugates attached to the membranes of cells in the upper respiratory tract and lungs (1), to facility its function.
HA is formed via the proteolytic cleavage of the precursor protein HA0 into either HA1 or HA2, and the inhibitors of HA can be classified into two different subtypes. The first type of inhibitor blocks the association between HA1 and the neuraminic acid (Neu) receptors on the surface of the target host cells. The second type of inhibitor, such as BMY-27709 (61, 62) and stachyflin (63-65), interrupts the HA2- mediated fusion process (Figure 7). This second type of inhibitor can inhibit the conformational transition of HA2 that is induced by lower pH values.
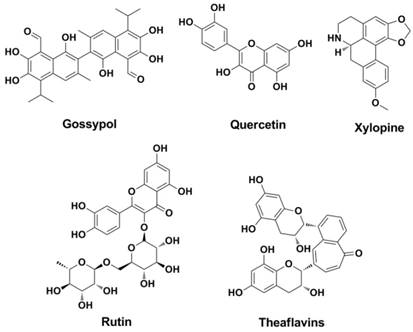
Extracted from cotton plant in 1970s, a natural phenolic aldehyde named Gossypol (Figure 6) was found to be effective against pneumonia caused by influenza virus (66-69). Further studies found that other natural products (or its derivatives) such as Rutin (70, 71), Quercetin (71-74), Xylopine (71, 75) and Theaflavins (76, 77) are HA inhibitors that interacts with HA (Figure 6).
Structures of N-acetylneuraminic acid and neuraminic acid.
Structures of typical natural products as HA inhibitors.
Structures of newly developed HA inhibitors.
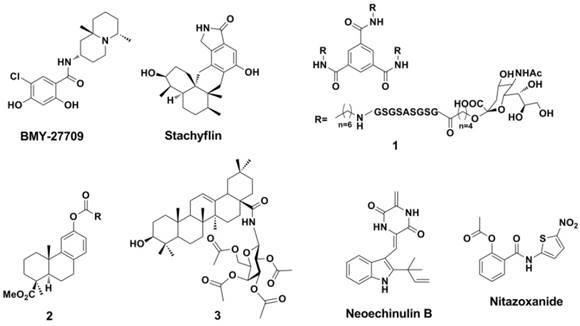
BMY-27709, contains a salicylamide scaffold and has been identified as specific to the influenza A virus (with IC50 values of 3-8 mM in a multicycle replication assay for A/WSN/33 virus) (61). BMY-27709 was found to inhibit the H1 and H2 viruses (though it is inactive against H3 virus) in the early stage of infection. Further study has indicated that BMY-27709 blocks the HA-mediated fusion process (62). Stachyflin (Figure 7) is a HA inhibitor that is similar to BMY-27709 in that it has activity against the H1 and H2 influenza A viruses (64). Experiments have suggested that Stachyflin blocks the HA-mediated cell fusion process by inhibiting the conformational transition of the HA protein (64, 65).
It is interesting that a trivalent glycopeptide mimetic (compound 1, Figure 7) displayed inhibitory activity against HA (H5) of avian influenza (inhibitory constant ( Ki ) = 15 μM and compound 1 is not a small molecular but this compound cannot simply be subdivided into peptide either) (2). The follow-up work carried out by Zhao et al found that a series of podocarpic acid derivatives (compound 2, Figure 7) exhibited potent activities (EC50 = 140-640 nM ) against a cell-line adapted influenza virus (A/Puerto Rico/8/34, PR8, an oseltamivir and amantadine resist H1N1 strain) infection of MDCK (Madin-Darby canine kidney) cells (78). Natural product pentacyclic triterpenoids (compound 3, Figure 7) exhibited inhibitory against influenza viruses, which were comparable to or even more potent than that of oseltamivir (79). Compound 3 was effective against the A/HuNan-ZhuHui/1222/2010 H3N2 strain (amantadine and ribavirin resistant), the A/LiaoNing-ZhenXing/ 1109/2010 H1N1 strain (oseltamivir-resistant), and even the influenza B/ShenZhen/155/2005, with EC50 values of 3.18, 6.58, and 2.80 μM (MDCK cell- based Cytopathic effect reduction assay, CPE Assay). Obtained from marine-derived fungus Eurotium rubrum, a class of novel prenylated indole diketopiperazine alkaloids displayed potent inhibition against H1N1 virus including oseltamivir and amantadine-resistant clinical isolates (80). The indole alkaloids (such as Neoechinulin B) can disrupt the interaction between the virus and host cells through binging to influenza envelope HA (80). The EC50 valuses of Neoechinulin B against A/LiaoNing-ZhenXing/1109/2010 H1N1, A/HuNan-ZhuHui/1222/2010 H3N2 and A/WSN/33 H1N1 were 16.89, 22.22 and 27.4 μM, respectively (MDCK cell- based CPE Assay).
Thiazolides such as Nitazoxanide (Figure 7) are powerful broad-spectrum antiviral drugs by blocking the maturation of viral HA, which made them be active against influenza viruses (81-86). The IC50 valuses of Nitazoxanide against influenza A/WSN/1933 H1N1, A/Parma/24/2009 H1N1 (Oseltamivir-resistant), A/Parma/06/2007 H3N2 (amantadine-resistant), A/goose/Italy/296246/2003 H5N9 and A/turkey/Italy/RA5563/1999 H7N1 were 1.6, 1.9, 1.0, 3.2 and 1.6 μM, respectively (MDCK cell-based CPE assay).
1.3 NA inhibitors (NAIs)
NA (also called sialidase) is a viral enzyme that is made up of four identical subunits and is anchored to the membrane of the virus (87). NA plays a key role in the spreading of the virus. The terminal neuraminic acid residues of the glycoproteins of the newly formed virion progeny form glycosidic linkages with the neuraminic acid receptor on the host-cell surface; this glycosidic linkage is cleaved by NA, which thereby assists in the release of the virion progeny from the infected cells (88, 89). Therefore NA is an attractive target for anti-influenza research, and inhibitors of NA containing a Neu core have attracted much attention (90-92).
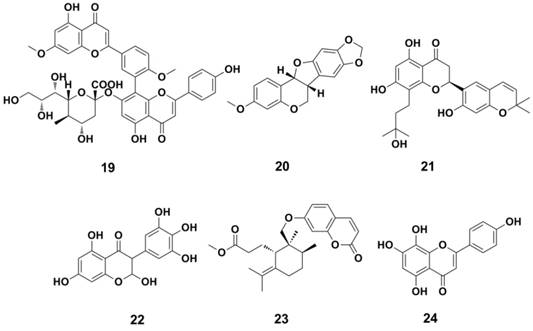
Meindl et al synthesized FANA (93, 94) and DANA (Neu5Ac2en)(94, 95), which are analogues of sialic acid (Figure 8). However, further study indicated that DANA has limited activity and failed as a clinical treatment of influenza (95-97). Von Itzstein and colleagues (98) modified the structure of DANA to synthesize the novel NA inhibitor Zanamivir (Figure 8). Zanamivir was effective against influenza A/(Singapore/1/57 and B/Victoria/102/85 virus with IC50 values of 14 nM and 5 nM, respectively (MDCK cell-based Plaque Reduction Assay). Although the oral bioavailability of Zanamivir is very low (2%-3%), the FDA approved it in 1999 as the first NA inhibitor agent (formulated for oral inhalation).
Based on the structure of Zanamivir and the 3-dimensional structure of Zanamivir and influenza-virus NA (subtype N9) (99) , Kim et al (100) synthesized Oseltamivir (Figure 8), which has enhanced oral bioavailability. The FDA also approved Oseltamivir in 1999, and it is the most popular NA inhibitor in the clinic at present. The activities of Oseltamivir carboxylate against representative N3-N9 NAs with IC50s range from 0.3 nM to 1.5 nM (Neuraminidase Enzyme Assay). More importantly, Zanamivir and Oseltamivir are effective against amantadane resistant strains.
Peramivir (101) and Laninamivir (102, 103) are also used as NA inhibitors and currently licensed in Asian countris (Figure 8). Peramivir is only administered intravenously because of its poor bioavailability (104). Laninamivir showed potent NA inhibitory activities against 11 strains of H1N1 viruses, 15 strains of H3N2, 23 strains of B viruses with IC50 valuses range 1.29-5.97 nM, 7.09-38.8 nM and 10.4-31.4 nM, respectively (Enzymatic assays). Laninamivir (also formulated for oral inhalation) is a long-term NA inhibitor, and it appears to be effective for patients with Oseltamivir resistance (94, 105-107). The suggested treatment usage of laninamivir (Japan) is beneficial when patients confer resistance to oseltamivir (94, 106, 107).
The recent progress in researching NA inhibitors has mainly focused on the structure modification/optimization of Zanamivir and Oseltamivir, both of which are similar to the Neu analogues (compounds 4-9, Figure 8) (108-115).
The development of irreversible inhibitors is an attractive strategy for overcoming drug resistance. Designed and synthesized by Kim et al. (116), those compounds (represented by compound 10, Figure 8) could form transient covalent intermediates with Tyr406 located at the catalytic domain of NA, thereby gaining potent broad-spectrum inhibitory activity against drug-resistant strains. Furthermore, those compounds exhibited equivalent or better drug efficacies in animal experiments.
Structures of representative NAIs.
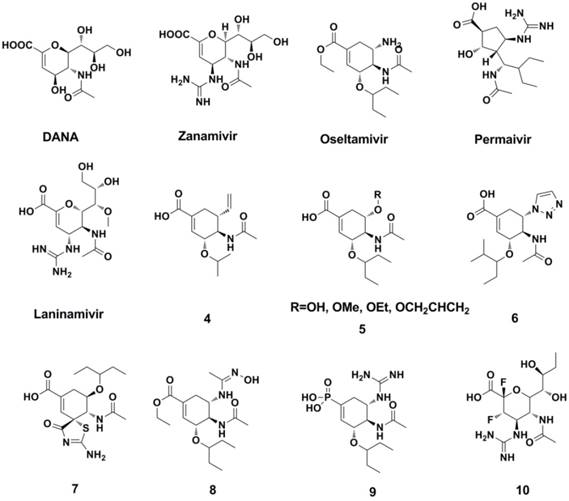
Structures of dimeric zanamivir conjugates and benzoic acid derivatives.
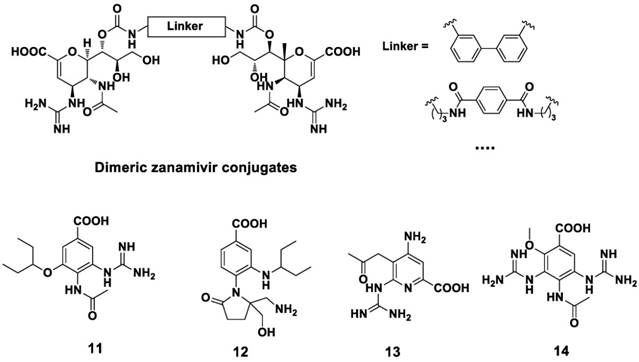
Structures of pyrrolidine derivatives as NAIs.
Remarkably, dimeric (or tethered) NA inhibitors developed by Tucker and co-workers are prospective for the clinical realization as anti-influenza agents (117-120). Dimeric zanamivir conjugates were synthesized and proved to be highly potent NA inhibitors (Figure 9) (120, 121). These dimeric compounds showed broad-spectrum activity and were 10-1000 fold more potent than that of zanamivir.
With the aim to find novel, potent NA inhibitor, benzoic acid derivatives (Figure 9) were developed in recent studies (122-125). However, the plane of the aromatic ring may limit the orientation of the substituent group, resulting in the poor activity of these benzoic acid derivatives. Pyrrolidine derivatives are also effective against the influenza A virus (Figure 10). Wang et al (126) found compound 15 showed anti-influenza activity ( IC50 = 0.2 μM against NA A/Tokyo and 8 μM against NA B/Memphis, NA inhibition assay). Compound 16 (127) and compound 17 (128), showed anti-influenza activity against influenza A and influenza B, respectively. Compound 18, as an analogue of L(-)-proline, showed inhibitory activity against NA from the influenza A virus (129).
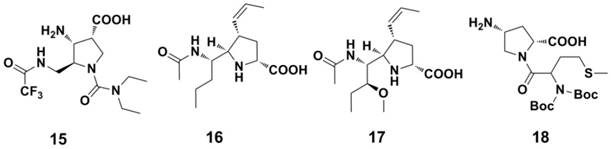
Some natural products have also been found to possess anti-influenza activities in past few years (Figure 11). Ginkgetin-sialic acid conjugates (compound 19) (130), significantly improved the survival rate of mice infected with the influenza virus. Flavanones and flavonoids (Figure 11, 20-23) also showed potent NA inhibition (IC50 ranges 1.4-20 μM against NA, NA inhibition assay) (131-134). Isoscutellarein (compound 24) and its derivatives are also active in cellular assays (EC50 = 20 μM against influenza A/Guizhou/54/89 H3N2, MDCK cell-based CPE assay) and animal models (135, 136).
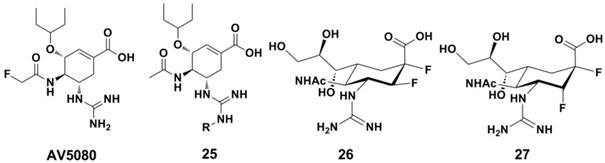
A novel highly potent oral drug candidate AV5080 (Figure 12) exhibited subnanomolar activity against influenza virus NA in vitro (with IC50s = 0.03 nM and 0.07 nM against NA of A/Duck/Minnesota/1525/1981 H5N1 and A/Perth/265/2009 H1N1 in NA enzyme based assays, respectively) (137). The N-substituted Oseltamivir analogues (compound 25, Figure 12) displayed enhanced inhibition against NA from Oseltamivir-resistant and wild-type strains (138). Jin-Hyo Kim et al synthesized a series covalent NA inhibitors (represented by compound 26 and 27, Figure 12) by introducing the strong electronegative fluorine atom at core-ring of Zanamivir and Oseltamivir (116); and these compounds showed excellent antiviral activity in vitro. Compound 27 showd IC50 values of 1 nM and 10 nM agaisnt B/Perth/211/01 and A/Fukui/45/01 H3N2 in plaque size reduction assays, superior than those for Zanamivir (10 nM and 100 nM, respectively). These compounds also showd comparable inhibition levels in animal models (compared with Zanamivir) (116).
1.4 RNA-dependent RNA polymerase (RdRp) inhibitors
The RNA-dependent RNA polymerase (RdRp) of the influenza virus has been highly conserved among all strains and subtypes during evolution (139). Unlike mammals, the RdRp of the influenza virus exhibits activities of both replicases and endonucleases. During the early stages of infection, RdRp synthesizes mRNA using vRNA as a template. During the advanced stage of infection, as the conformation of RdRp changes, RdRp becomes responsible for the catalytic synthesis of cRNA and vRNA (140-143). RdRp was also named “3P-complex” because it is composed of three subunits (PA, PB1 and PB2). RdRp plays a critical role during the life cycle of the virus. Therefore, it has become a promising target for the development of anti-influenza drugs in recent years (144, 145). Based on the mechanism of the interactions berween inhibitors and polymerase, RdRp inhibitors can be easily subdivided into the following four subtypes (146, 147): (i) RdRp disrupting compounds, (ii) PB2 cab-binding (PB2-CBD) inhibitors, (iii) PA endonuclease inhibitors, (iv) PB1 or nucleoside analogue like inhibitors.
RdRp disrupting compounds: An attractive strategy for developing RdRp inhibitors appears to be interrupting the subunits interactions (the assembly course of the subunits in to a functional polymerase complex), and this strategy proves to be effective in recent studies. Figure 13 shows the structures of typical RdRP disrupting inhibitors (compounds 28-33, Figure 13) (17, 148-153), and RdRp disrupting compounds also named prpotein-protein interaction inhibitors (PPI inhibitors) because of its interference/or inhibiton of the protein-protein interaction in the assembly course.
Structures of nature products as NAIs.
Structures of newly developed NAIs.
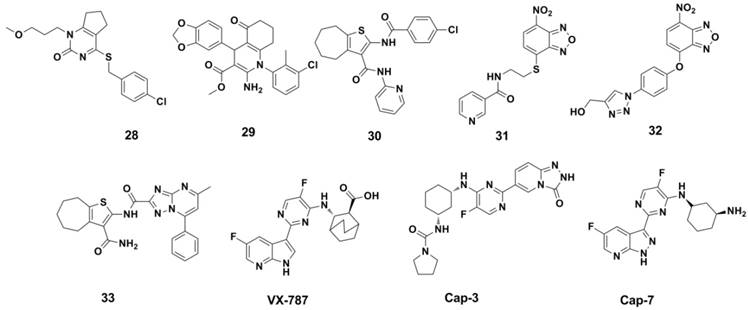
PB2 cab-binding inhibitors: The cap-binding activity resides in PB2 subunit was discovered more than 30 years ago (154, 155), but the binding mode of influenza A and B was revealed since the X-ray structure of the PB2 cap-binding domain was reported in 2008 (156). PB2-CBD as a drug target was validated by the clinical candidate VX-787 (94, 157-159) (Figure 14). As an azaindole based inhibitors, VX-787 was able to occupy the m7GTP binding pocket in the PB2-CBD of influeza A (demonstrated by X-ray structure) (156). VX-787 displays potent antiviral activity against a widely range of influenza A virus strains in cellular assays (with EC50s in nanomolar range), including amantadine- and NAI-resistant strains. Cap-3 and Cap-7 (Figure 14) were also cap-binding inhibitors and were reported recently by Roch et al (160). These two compounds can inhibit the transcription process in enzymatic assay and inhibit the virus replication process in cell experiments (with EC50s range 1-9 μM).
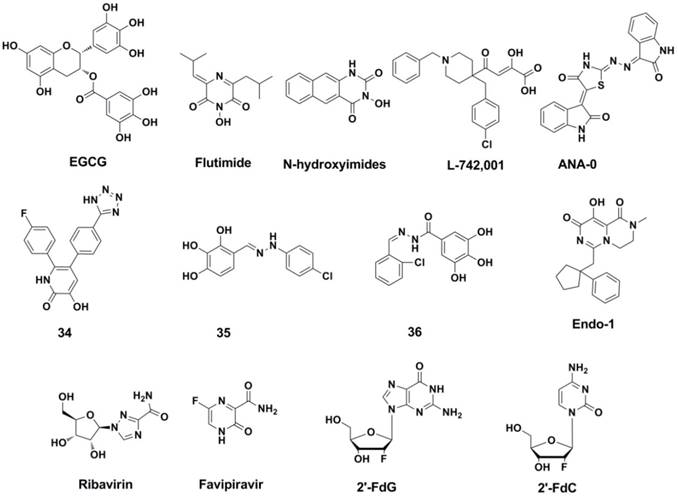
PA endonuclease inhibitors: Another promising target in polymerase may be the conserved residues inside of the catalytic site of N-terminal domain of PA. This stragety was also validated since the introduction of AL-794 and S-033188 (structures undisclosed) as PA inhibitors into clinical trials (161). The challenge of the strategy is to achieve inhibitors consist of metal-chelating scaffolds to bind the divalent metal ion(s), and occupy the PA-Nter catalytic site. Many inhibitors of this type have been report so far, and these inhibitors have in common that they possess chelating motifs (147). These inhibitors include: EGCG (162, 163) and its aliphatic analogues (EGCG analogues also showed inhibitory activity against Neuraminidase) (164), N-hydroxamic acids and N-hydroxyimides (165), flutimide (166) and its aromatic analogues (167), tetramic acid derivatives (168), L-742,001 (169, 170), ANA-0 (171), polyphenolic catechins (162), phenethyl-phenylphthalimide analogues (172), macrocyclic bisbibenzyls (163, 173), pyrimidinoles (174), fullerenes (175), hydroxy- quinolinones (176), hydroxypyridinones (IC50 = 11 nM in Enzymic Assay, compound 34) (177), hydroxypyridazinones (178), trihydroxy-phenyl-bearing compounds (compound 35 and 36) (179-181), 2-hydroxy-benzamides(182), hydroxy-pyrimidinones (183), β-diketo acid and its bioisosteric compounds (184), thiosemicarbazones (183), bisdihydroxyindole-carboxamides (185), pyrido-piperazinediones (Endo-1) (160) and miscellaneous compounds (186). However, quite a number of these compounds showed in cell experiments, which is likely to be connected with the poor cellular uptake and/or insufficient anti-viral activity and selectivity. Structures of the representative inhibitors were shown in Figure 14.
Nucleoside and nucleobase analogue inhibitors: This subtype of RdRp inhibitors is likely to be most promising in anti-influenza drug development because of the following advances: low cytotoxicity, high resistance barrier and broad coverage of diverse RNA viruses (147).
Ribavirin (Figure 14) (187) and Favipiravir (188, 189) (also named T-705, Figure 14) are RdRp inhibitors that both have a nucleoside fragment. Ribavirin was approved as a broad-spectrum antiviral drug for years (190, 191) and Favipiravir has advanced to phase II clinical trials (USA) and phase III clinical trials (Japan). Ribavirin can influence the DNA/RNA synthesis of the host cell (192, 193) through a combination of several different mechanisms (147). Further research reveals that ribavirin and its analogues (5-azacytidine and 5-fluorouracil) are lethal mutagens of influenza virus (194).
Structures of RdRp disrupting compounds and PB2 cab-binding inhibitors.
Structures of representative PA endonuclease inhibitor, Nucleoside and nucleobase analogue inhibitors.
As a nucleobase mimetic, Favipiravir and its analogues showed to be effective against strains that are resistant to NA inhibitors and M2 ion channel protein inhibitors (188, 195-199).
2ʹ-Deoxy-2ʹ-fluoroguanosine (2'-FdG) (200) and other nucleoside analogues (such as C-3ʹ-modified analogues (201), 2ʹ-substituted carba-nucleoside analogues (202), 6-methyl-7-substituted-7-deaza purine nucleoside analogues (203) etc.) were reported to posess anti-influenza activities against influenza A and B viruses. 2'-FdG can inhibit the polymerase complex through nonobligate chain termination. As a pyrimidine analogue of 2'-FdG, 2ʹ-deoxy-2ʹ-fluorocytidine (2'-FdC) seems more potent against various strains of influenza A and/or B in vitro and in vivo (204). Although these nucleoside analogues showed strong inhibitory activities, the clinical application was limited because the therapeutic window is too narrow (147).
1.5 Nucleocapsid protein (NP) inhibitors
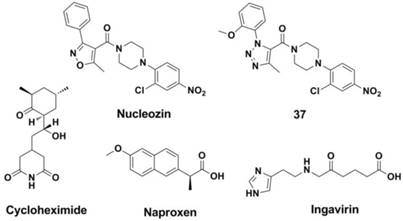
NPs account for 30% of the total protein of the virus and, as a structural protein, form the virus ribonucleoprotein (vRNP) (205, 206). NP has multiple functions in the virus and is involved in replication, the formation of specificity to the host, and other activities (7, 207). Recent research has resulted in the discovery of some potential targets and inhibitors of NP (7, 148, 208-210). A study carried out by Ye and co-workers indicated that the tail loop-binding pocket in the influenza A virus NP could be a potential site for antiviral development. Kao et al (210) found a small molecular, nucleozin (NCZ, Figure 15), which may inhibit the infection caused by the H1N1, H3N2 and H5N1 strains as it can initiate the aggregation process of NP, blocking its nuclear accumulation (with EC50s of 0.069 μM, 0.16 μM and 0.33 μM showd in MDCK cell-based Plaque Reduction Assay, respectively). Their work also proved that viral NP is a potential target for the development of anti-influenza drugs. Ke ding et al (211) replaced the isoxazole ring of nucleozin with triazole to obtain compound 37 (Figure 15), which showed enhanced activity against the replication of various H1N1, H3N2, H5N1 and H9N2 influenza A virus strains (with IC50s ranges 0.15-12.4 μM, MDCK cell-based Anti-influenza Assay). Furthermore, compound 37 was also effective against strains that are resistant to amantadine (A/WSN/33, H1N1) and oseltamivir (A/WSN/1933 H1N1, 274Y).
Small molecules such as Cycloheximide (CHX, Figure 15) (212-214) and Naproxen (Figure 15) (215) were found to be effective against the functional polymerization of the NP monomers. Furthermore, a licensed drug named Ingavirin (approved in Russia, Figure 15) interacts with NP directly by interrupt transportation of newly synthesized NPs to the nucleus (173, 216-221).
Structures of NP inhibitors.
1.6 M2 ion channel inhibitors
The M2 ion channel protein is a transmembrane protein that possesses the activities of typical ion channels (222-225). Influenza B lacks an M2 ion channel protein, but the B/M2 protein functions as an M2 ion channel protein during the assembly of the virus (225). The M2 and B/M2 proteins play important roles in the incorporation of the viral ribonucleoprotein (vRNP) complex into the virus during the assembly process.
Although the M2 inhibitors (Amantadine and Rimantadine) are firstly approved and recommend for use in clinical, but drug resistance has limited their clinical use. Most H1N1, H3N2 strains and virus B are resistant to Amantadanes, but the resistance to Zanamivir and Oseltamivir is very low. Furthermore, neurotoxic effects (such as confusion, disorientation, anxiety, jitteriness, etc.) caused by Amantadine are usually more common when the drug is used more than a week. Since the structure of M2 channel protein has been determined (226), developing more potent drugs against H1N1 influenza virus and solving the drug-resistant problem become promising (226-229).
M2 inhibitors can be classified as two groups based on the structure of the inhibitors (34). The first group includes Amantadine and Rimantadine (225) analogues (226, 230-234) (Figure 16, compounds 38-40). Amantadine and compound 39 showed activities against influenza A H3N2 virus with IC50s of 3.35 and 8.58 μM in a MDCK cell-based CPE Assay, respectively. The second group is non-adamantane derivatives and promising drugs (235, 236). Polyamines are effective against influenza virus (such as Spermine and Spermidine, Figure 16) (237, 238). This is because there is another binding site for polyamines at the M2 protein (significantly distinct from the Amantadine binding site) (239), and polyamines have attracted much attention for developing novel anti-influenza drugs (240, 241). Spiropiperidine and its derivatives (represented by compound 41 in Figure 16) are also effective against M2 protein and showed activities against amantadine-resistant viruses (34). Nature products such as pinanamine derivatives (compound 42 in Figure 16) also exhibit good anti-influenza activities (242). However, almost none of M2 inhibitors are effective against the influenza B virus (225).
Structures of M2 ion channel inhibitors.
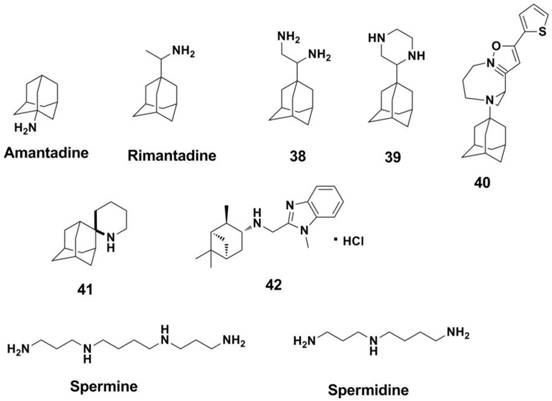
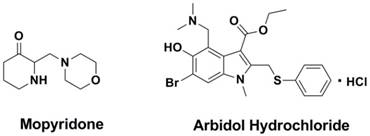
Mopyridone (Figure 17), a compound which was screened from a series of tetrahydro-2(1H)-pyrimidinone derivatives by Galabov et al in 1980s (243, 244), demonstrated to be a large scope anti-flu effect and large spectrum of influenza A1H1, AH2N2, AH3N3 and B strains in vitro and in vivo (245). What is particularly noteworthy is that this compound is the only anti-flu compound for which it was proved to have as a target M1 protein (246), and further research shows this compound with low acute toxicity in mice (247).
1.7 Arbidol hydrochloride
Developed by the Pharmaceutical Chemistry Research Center of the former Soviet union (248-251), arbidol hydrochloride (AH, Figure 17) was selected as a new antiviral drug for influenza infection in Russia and China (250, 251). AH is effective for the prophylaxis and treatment of influenza and other acute respiratory viral infections (ARVI) (250). AH inhibits viral entry into target cells and also stimulates the immune response (252). At the same time, AH has immunomodulatory activity, a capacity to induce interferons, and antioxidant properties. Although some studies from Russia and China have proved AH to be effective, it has not been approved for use in western countries.
Structures of Mopyridone and Arbidol Hydrochloride.
2. Agents targeted at potential host sites
The potential targets for antiviral therapies include proteases, vacuolar-type proton-adenosine triphosphatases (V-ATPases), kinases, and other proteins. However, the effectiveness of inhibitors against these enzymes needs further improvements.
2.1 Protease inhibitors
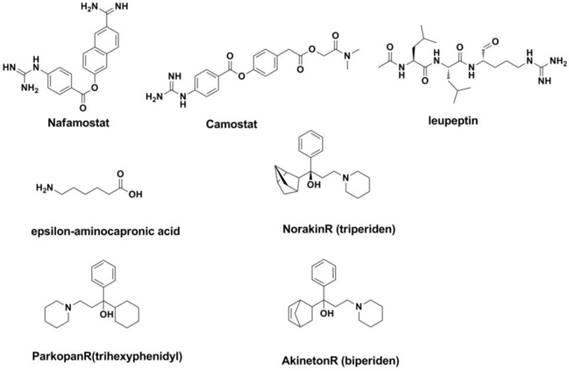
The interaction between the host protease and the splice site of the viral precursor protein HA0 determines whether the species is infected with influenza and affects the virulence of the virus. For example, the splice site of HA0 in highly pathogenic avian influenza strains such as H5 and H7 can be easily be spliced by the widespread basic amino acid protease or PC6 serine protease, resulting in fatal systemic infection in birds (253, 254). In fact, known inhibitors such as nafamostat, Leupeptin (255), epsilon-aminocapronic acid (256), Camostat (257), Aprotinin (258) have been studied for years (Figure 18). Some of them (such as Camostat) have shown selective inhibition against the influenza A and influenza B viruses in vitro and in vivo (259).
2.2 V-ATPase inhibitors
The selective V-ATPase inhibitors may increase the internal pH of the prelysosome, thereby inhibiting the conformational transformation of HA from the unfused conformation to the fused conformation. This would result in the inhibition of replication during the course of the viral life cycle. It is interesting that four drugs used in the treatment of Parkinson's disease (NorakinR, ParkopanR, AntiparkinR and AkinetonR) all contain an adamantine scaffold that has shown inhibitory activity against the influenza A and influenza B viruses (260-262).
Structures of agents targeted at potential sites of host.
2.3 Anti-oxidants
Increasing evidence has indicated that the oxidation plays a major role in influenza virus life cycle and replication.
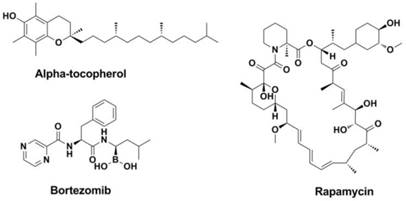
Among the antiviral strategies make use of anti-oxidants, alpha-tocopherol (Figure 19) enjoys a long history since 1960s (263-266). Further researches revealed that alpha-tocopherol (or combination) could normalize the lipid peroxidation processes caused by viral infection (264, 267-272).
The activation of the NADPH oxidase 2 (NOX2) might promote the respiratory symptoms result from infection with influenza A viruses and impede the clearance of the virus (273-277). Therefore, NADPH oxidases became promising novel pharmacologic targets against influenza A virus infection (278-280).
Structures of alpha-tocopherol, Rapamycin and Bortezomib.
3. Other targets and strategies
As described above, the research into the development of anti-influenza drugs has been ongoing for decades and has greatly progressed. However, the drug resistance acquired in recent years from the widespread use of anti-influenza drugs has prompted researchers to find new potential targets.
Pathway inhibitors: The mTOR inhibitor Rapamycin (Figure 19), which has been marketed as an immunosuppressive drug that surprisingly leads to the protection from infection with multiple subtypes of the influenza virus (6). In addition, some proteasome inhibitors, such as NFKβ inhibitors (Bortezomib, Figure 19), Raf/MEK/ERK pathway inhibitors are also effective and can work as new antivirals against influenza virus (281).
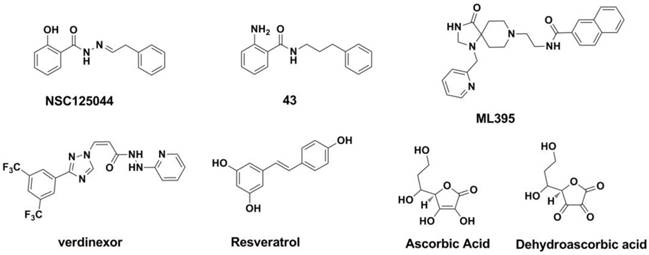
NS1 inhibitors: The non-structural protein 1 (NS1A) of the influenza virus is a small, multifunctional protein that plays a critical role in the response of the host antiviral process (282, 283). When NS1A binds to cleavage and polyadenylation specificity factor (CPSF30), the maturation of the host RNAs is blocked, which leads to the reduction of host proteins (282-286). Twu and co-workers (287) found that the binding of CPSF30 is mediated by the second and third zinc fingers (F2F3) of CPSF30. When the binding process of CPSF30 to the NS1A protein was blocked by a fragment which containing the F2F3 binding motif, the replication of the influenza A virus was inhibited. This work indicated that the CPSF30 binding site in the NS1A protein is a potential target for antiviral therapies against the influenza A virus (287). A recent study carried out by Jablonski et al. showed a series of compounds derived from the NSC125044 (Figure 20, compound 43) displayed inhibitory activity against NS1 protein (288).
Phospholipase inhibitors: Phospholipase D (PLD) is one kind of phospholipase that catalyzes the formation of phosphatidic acid, an important messenger in signaling and metabolic pathways (289). Recent studies show that human PLD2 inhibitor such as ML395 (Figure 20) possess a broad-spectrum inhibitory against influenza strains (290).
vRNPs inhibitors: As the exportion of influenza virus vRNPs from nuclear has been demonstrated to be mediated by hostexportin 1 (XPO1) (291-293), developing inhibitors of XPO1 to interrupt the vRNP exportion process and then hinder the replicationcycle of the virus make some sense. A study performed by Olivia Perwitasari et al. show that verdinexor (Figure 20), a novel selective inhibitor of XPO1 selectively and potently inhibited the replication process of various influenza virus A and B strains in vitro (294). Resveratrol (Figure 20) could interrupt the translocation process of RNPs from the nucleus to the cytoplasm and may be useful as an anti-influenza drug (295-297). Ascorbic and dehydroascorbic acids (Figure 20) also possess the antiviral effect, and this effect may work at the envelopment of viral nucleocapsids after the completion of viral DNA replication (298, 299).
RNA inhibitors: RNA inhibitors refer molecule that regulate the expression of gene. RNA inhibitors play important roles in RNA silencing, RNA interference (33) and post-transcriptional regulation of gene expression. RNA inhibitors are able to regulate the expression of influenza viral RNAs. Accordingly, viral genes became a potential target and RNA inhibitors may be effective in influenza treatment (300).
Immunomodulatory agents: Inflammatory changes and other immune reactions that associated with acute coronary syndrome may influence the mortality of influenza (301). Immunomodulatory agents can reduce levels of LDL-cholesterol and improve the inflammatory changes. Studies show that statin treatment in pneumonia patients or influenza patients exhibited reduced mortality (301-304). Other immunomodulatory agents (305) such as Cyclooxygenase inhibitors (aspirin) (306), ACE inhibitors (ACEIs) (302, 307), angiotensin receptor blockers (ARBs) (302), AMPK agonists (metformin) (308), PPARα and PPARγ agonists (fibrates and glitazones) (309-311), however, showd the ability to reduce mortality in mouse models of influenza (312, 313) and patients with pneumonia (ARBs and ACEIs) (302, 314).
Combination therapies: Combination therapy is one of the prospective domains in the investigation for anti-flu agents. Combination therapies used in anti-influenza treatment may improve the clinical outcomes and enhance antiviral activity against drug-resistant strains. They can also reduce the risk of side effects, dose-related toxicity, mortality and morbidity (315-317). Therefore, combination therapies are recommend in clinical and can be classified in to early combination chemotherapy and sequential multidrug chemotherapy. A classical combination is M2 blockers and NA inhibitors to avoid drug-resistance. Many studies have been carried out to evaluate the efficacy of combination therapies and single-drug treatment, and most combination therapies showed superior outcomes in mice models (272, 318-321).
Conclusion
As drug resistance (often caused by mono-therapy and, sometimes, uncontrolled use in farm animals) (322) and frequent mutations of strains are increasingly serious in the past few years, few drugs can be effective in this situation. The development of antiviral agents is a practical significance topic that has attracted much attention and had made great progress during the past several decades. Small molecular inhibitors are powerful weapon to fight against influenza virus. Small molecular inhibitors function as M2 ion-channel inhibitors, NA inhibitors and protease inhibitors are used in clinical. M2 ion-channel inhibitors were firstly used in clinic but they have shown some defects in clinical use. The quick development of drug-resistance (strains such as H1N1, H3N2 and type B viruses) has limited their clinical use. Alternatively, NA inhibitors are currently the most popular targets of antiviral research (Oseltamivir and Zanamivir are effective against amantadane resistant strains). While arbidol hydrochloride is effective against the influenza A and B viruses, its precise mechanisms of action remain unclear. The usefulness of the marketed NA inhibitors oseltamivir and Zanamivir is also limited due to the increasing prevalence of resistant strains. Besides mono-therapy, combination therapies were also developed (29, 315, 317). Treatment with immunomodulatory agents also represents a new approach to deal with seasonal and pandemic influenza. Along with the determination of the NA structure and the discovery of SAR in recent years, the basis of rational design for novel, potent NAIs is valid. As drug resistance became an increasingly serious problem, and the existing drugs may no longer be useful due to the emergence of mutated strains, researchers have focused on making structural modifications of current drugs to identify potential new targets such as HA (HA) inhibitors, RNA-dependent RNA polymerase (RdRp) inhibitors, nucleocapsid protein (NP) inhibitors, protease inhibitors, V-ATPase inhibitors, Pathway inhibitors, Anti-oxidants, Phospholipase inhibitors, vRNPs inhibitors and RNA inhibitors etc. Some inhibitors of these targets have shown certain anti-influenza activities in vivo and in vitro, and further studies are in progress. It is particularly worth mentioning here that major progress was made in unravelling of functioning and mechanisms of the virus polymerase in past few years, as well as novel RdRp inhibitors. It can be anticipated that polymerase inhibitors will reshape the field of influenza prevention and therapy in the near future. There is, however, still a long way to go in winning the battle against influenza pandemics.
Structures of NS1 inhibitors and phospholipase inhibitors.
Acknowledgements
This study was supported by the National Natural Science Foundation of China (Grant Nos. 81571637, 81271532 and 81101079) and the Fundamental Research Funds for the Central Universities (Project No. 2015SCU04B09).
Disclosure
The authors declare no conflict of interest.
References
1. Wagner R, Matrosovich M, Klenk HD. Functional balance between haemagglutinin and neuraminidase in influenza virus infections. Rev Med Virol. 2002;12:159-66
2. Waldmann M, Jirmann R, Hoelscher K, Wienke M, Niemeyer FC, Rehders D. et al. A nanomolar multivalent ligand as entry inhibitor of the hemagglutinin of avian influenza. J Am Chem Soc. 2014;136:783-8
3. Lam TT, Wang J, Shen Y, Zhou B, Duan L, Cheung CL. et al. The genesis and source of the H7N9 influenza viruses causing human infections in China. Nature. 2013;502:241-4
4. World Health Organization Influenza Fact Sheet 211. In: Centre M, editor. World Health Organization. Geneva: World Health Organization. 2014
5. von Itzstein M. The war against influenza: discovery and development of sialidase inhibitors. Nat Rev Drug Discov. 2007;6:967-74
6. McMichael AJ, Haynes BF. Influenza vaccines: mTOR inhibition surprisingly leads to protection. Nat Immunol. 2013;14:1205-7
7. Das K. Antivirals targeting influenza A virus. J Med Chem. 2012;55:6263-77
8. Dreyfus P. [Treatment of influenzal infections by a moroxydine derivative]. Sem Ther. 1966;42:51-2
9. Oxford JS. Influenza A pandemics of the 20th century with special reference to 1918: virology, pathology and epidemiology. Rev Med Virol. 2000;10:119-33
10. Jagger BW, Wise HM, Kash JC, Walters KA, Wills NM, Xiao YL. et al. An overlapping protein-coding region in influenza A virus segment 3 modulates the host response. Science. 2012;337:199-204
11. Medina RA, Garcia-Sastre A. Influenza A viruses: new research developments. Nat Rev Microbiol. 2011;9:590-603
12. Chen W, Calvo PA, Malide D, Gibbs J, Schubert U, Bacik I. et al. A novel influenza A virus mitochondrial protein that induces cell death. Nat Med. 2001;7:1306-12
13. Wise HM, Foeglein A, Sun J, Dalton RM, Patel S, Howard W. et al. A complicated message: Identification of a novel PB1-related protein translated from influenza A virus segment 2 mRNA. J Virol. 2009;83:8021-31
14. Muramoto Y, Noda T, Kawakami E, Akkina R, Kawaoka Y. Identification of novel influenza A virus proteins translated from PA mRNA. J Virol. 2013;87:2455-62
15. Tong S, Zhu X, Li Y, Shi M, Zhang J, Bourgeois M. et al. New world bats harbor diverse influenza A viruses. PLoS Pathog. 2013;9:e1003657
16. Das K, Aramini JM, Ma LC, Krug RM, Arnold E. Structures of influenza A proteins and insights into antiviral drug targets. Nat Struct Mol Biol. 2010;17:530-8
17. Loregian A, Coen DM. Selective anti-cytomegalovirus compounds discovered by screening for inhibitors of subunit interactions of the viral polymerase. Chem Biol. 2006;13:191-200
18. Jones JC, Turpin EA, Bultmann H, Brandt CR, Schultz-Cherry S. Inhibition of influenza virus infection by a novel antiviral peptide that targets viral attachment to cells. J Virol. 2006;80:11960-7
19. Altmann SE, Brandt CR, Jahrling PB, Blaney JE. Antiviral activity of the EB peptide against zoonotic poxviruses. Virol J. 2012;9:6
20. Nicol MQ, Ligertwood Y, Bacon MN, Dutia BM, Nash AA. A novel family of peptides with potent activity against influenza A viruses. J Gen Virol. 2012;93:980-6
21. Rajik M, Jahanshiri F, Omar AR, Ideris A, Hassan SS, Yusoff K. Identification and characterisation of a novel anti-viral peptide against avian influenza virus H9N2. Virol J. 2009;6:74
22. Matsubara T. Potential of peptides as inhibitors and mimotopes: selection of carbohydrate-mimetic peptides from phage display libraries. Journal of nucleic acids. 2012;2012:740982
23. Naganuma A, Mizuma H, Doi I, Ono A, Tsukioka G, Watanabe Y. et al. [A case of acute respiratory distress syndrome induced by fulminant influenza A (H3 N2) pneumonia]. Nihon Kokyuki Gakkai Zasshi. 2000;38:783-7
24. Yuan S. Drugs to cure avian influenza infection-multiple ways to prevent cell death. Cell Death Dis. 2013;4:e835
25. Leng YX, Yang SG, Song YH, Zhu X, Yao GQ. Ulinastatin for acute lung injury and acute respiratory distress syndrome: A systematic review and meta-analysis. World journal of critical care medicine. 2014;3:34-41
26. Belser JA, Lu X, Szretter KJ, Jin X, Aschenbrenner LM, Lee A. et al. DAS181, a novel sialidase fusion protein, protects mice from lethal avian influenza H5N1 virus infection. J Infect Dis. 2007;196:1493-9
27. Triana-Baltzer GB, Gubareva LV, Nicholls JM, Pearce MB, Mishin VP, Belser JA. et al. Novel pandemic influenza A(H1N1) viruses are potently inhibited by DAS181, a sialidase fusion protein. PLoS One. 2009;4:e7788
28. Zenilman JM, Fuchs EJ, Hendrix CW, Radebaugh C, Jurao R, Nayak SU. et al. Phase 1 clinical trials of DAS181, an inhaled sialidase, in healthy adults. Antiviral Res. 2015;123:114-9
29. Prabakaran M, Prabhu N, He F, Hongliang Q, Ho HT, Qiang J. et al. Combination therapy using chimeric monoclonal antibodies protects mice from lethal H5N1 infection and prevents formation of escape mutants. PLoS One. 2009;4:e5672
30. Xiang DX, Chen Q, Pang L, Zheng CL. Inhibitory effects of silver nanoparticles on H1N1 influenza A virus in vitro. J Virol Methods. 2011;178:137-42
31. Xiang D, Zheng Y, Duan W, Li X, Yin J, Shigdar S. et al. Inhibition of A/Human/Hubei/3/2005 (H3N2) influenza virus infection by silver nanoparticles in vitro and in vivo. International journal of nanomedicine. 2013;8:4103-13
32. McKenzie Z, Kendall M, Mackay RM, Tetley TD, Morgan C, Griffiths M. et al. Nanoparticles modulate surfactant protein A and D mediated protection against influenza A infection in vitro. Philos Trans R Soc Lond B Biol Sci. 2015;370:20140049
33. Torrecilla J, Rodriguez-Gascon A, Solinis MA, del Pozo-Rodriguez A. Lipid nanoparticles as carriers for RNAi against viral infections: current status and future perspectives. BioMed research international. 2014;2014:161794
34. Fytas C, Kolocouris A, Fytas G, Zoidis G, Valmas C, Basler CF. Influence of an additional amino group on the potency of aminoadamantanes against influenza virus A. II - Synthesis of spiropiperazines and in vitro activity against influenza A H3N2 virus. Bioorg Chem. 2010;38:247-51
35. Pu JY, He L, Wu SY, Zhang P, Huang X. [Anti-virus research of triterpenoids in licorice]. Bing Du Xue Bao. 2013;29:673-9
36. Kazakova OB, Giniyatullina GV, Yamansarov EY, Tolstikov GA. Betulin and ursolic acid synthetic derivatives as inhibitors of Papilloma virus. Bioorg Med Chem Lett. 2010;20:4088-90
37. Kazakova OB, Medvedeva NI, Baikova IP, Tolstikov GA, Lopatina TV, Iunusov MS. et al. [Synthesis of triterpenoid acylates - an effective reproduction inhibitors of influenza A (H1N1) and papilloma viruses]. Bioorg Khim. 2010;36:841-8
38. Flekhter OB, Medvedeva NI, Kukovinets OS, Spirikhin LV, Galkin EG, Galin FZ. et al. [Synthesis and antiviral activity of lupane triterpenoids with modified cycle E]. Bioorg Khim. 2007;33:629-34
39. Baltina LA, Flekhter OB, Nigmatullina LR, Boreko EI, Pavlova NI, Nikolaeva SN. et al. Lupane triterpenes and derivatives with antiviral activity. Bioorg Med Chem Lett. 2003;13:3549-52
40. Song G, Yang S, Zhang W, Cao Y, Wang P, Ding N. et al. Discovery of the first series of small molecule H5N1 entry inhibitors. J Med Chem. 2009;52:7368-71
41. Jia W, Wang C, Wang Y, Pan G, Jiang M, Li Z. et al. Qualitative and quantitative analysis of the major constituents in Chinese medical preparation Lianhua-Qingwen capsule by UPLC-DAD-QTOF-MS. TheScientificWorldJournal. 2015;2015:731765
42. Fiore C, Eisenhut M, Krausse R, Ragazzi E, Pellati D, Armanini D. et al. Antiviral effects of Glycyrrhiza species. Phytother Res. 2008;22:141-8
43. Harada S. The broad anti-viral agent glycyrrhizin directly modulates the fluidity of plasma membrane and HIV-1 envelope. Biochem J. 2005;392:191-9
44. Song W, Si L, Ji S, Wang H, Fang XM, Yu LY. et al. Uralsaponins M-Y, antiviral triterpenoid saponins from the roots of Glycyrrhiza uralensis. J Nat Prod. 2014;77:1632-43
45. Krumbiegel M, Dimitrov DS, Puri A, Blumenthal R. Dextran sulfate inhibits fusion of influenza virus and cells expressing influenza hemagglutinin with red blood cells. Biochim Biophys Acta. 1992;1110:158-64
46. Herrmann A, Korte T, Arnold K, Hillebrecht B. The influence of dextran sulfate on influenza A virus fusion with erythrocyte membranes. Antiviral Res. 1992;19:295-311
47. Yamada H, Moriishi E, Haredy AM, Takenaka N, Mori Y, Yamanishi K. et al. Influenza virus neuraminidase contributes to the dextran sulfate-dependent suppressive replication of some influenza A virus strains. Antiviral Res. 2012;96:344-52
48. Blaising J, Levy PL, Gondeau C, Phelip C, Varbanov M, Teissier E. et al. Silibinin inhibits hepatitis C virus entry into hepatocytes by hindering clathrin-dependent trafficking. Cell Microbiol. 2013;15:1866-82
49. Aggarwal BB, Deb L, Prasad S. Curcumin differs from tetrahydrocurcumin for molecular targets, signaling pathways and cellular responses. Molecules. 2015;20:185-205
50. Chen TY, Chen DY, Wen HW, Ou JL, Chiou SS, Chen JM. et al. Inhibition of enveloped viruses infectivity by curcumin. PLoS One. 2013;8:e62482
51. Shuto T. [Regulation of expression, function, and inflammatory responses of innate immune receptor Toll-like receptor-2 (TLR2) during inflammatory responses against infection]. Yakugaku Zasshi. 2013;133:1401-9
52. Guinea R, Carrasco L. Concanamycin A blocks influenza virus entry into cells under acidic conditions. FEBS Lett. 1994;349:327-30
53. Guinea R, Carrasco L. Concanamycin A: a powerful inhibitor of enveloped animal-virus entry into cells. Biochem Biophys Res Commun. 1994;201:1270-8
54. Yeganeh B, Ghavami S, Kroeker AL, Mahood TH, Stelmack GL, Klonisch T. et al. Suppression of influenza A virus replication in human lung epithelial cells by noncytotoxic concentrations bafilomycin A1. Am J Physiol Lung Cell Mol Physiol. 2015;308:L270-86
55. Ochiai H, Sakai S, Hirabayashi T, Shimizu Y, Terasawa K. Inhibitory effect of bafilomycin A1, a specific inhibitor of vacuolar-type proton pump, on the growth of influenza A and B viruses in MDCK cells. Antiviral Res. 1995;27:425-30
56. Di Trani L, Savarino A, Campitelli L, Norelli S, Puzelli S, D'Ostilio D. et al. Different pH requirements are associated with divergent inhibitory effects of chloroquine on human and avian influenza A viruses. Virol J. 2007;4:39
57. Paton NI, Lee L, Xu Y, Ooi EE, Cheung YB, Archuleta S. et al. Chloroquine for influenza prevention: a randomised, double-blind, placebo controlled trial. Lancet Infect Dis. 2011;11:677-83
58. Couceiro JN, Paulson JC, Baum LG. Influenza virus strains selectively recognize sialyloligosaccharides on human respiratory epithelium; the role of the host cell in selection of hemagglutinin receptor specificity. Virus Res. 1993;29:155-65
59. Skehel JJ, Wiley DC. Receptor binding and membrane fusion in virus entry: the influenza hemagglutinin. Annu Rev Biochem. 2000;69:531-69
60. Suzuki Y, Ito T, Suzuki T, Holland RE Jr, Chambers TM, Kiso M. et al. Sialic acid species as a determinant of the host range of influenza A viruses. J Virol. 2000;74:11825-31
61. Luo G, Colonno R, Krystal M. Characterization of a hemagglutinin-specific inhibitor of influenza A virus. Virology. 1996;226:66-76
62. Luo G, Torri A, Harte WE, Danetz S, Cianci C, Tiley L. et al. Molecular mechanism underlying the action of a novel fusion inhibitor of influenza A virus. J Virol. 1997;71:4062-70
63. Yoshimoto J, Kakui M, Iwasaki H, Sugimoto H, Fujiwara T, Hattori N. Identification of amino acids of influenza virus HA responsible for resistance to a fusion inhibitor, Stachyflin. Microbiol Immunol. 2000;44:677-85
64. Yoshimoto J, Kakui M, Iwasaki H, Fujiwara T, Sugimoto H, Hattori N. Identification of a novel HA conformational change inhibitor of human influenza virus. Arch Virol. 1999;144:865-78
65. Yagi S, Ono J, Yoshimoto J, Sugita K, Hattori N, Fujioka T. et al. Development of anti-influenza virus drugs I: improvement of oral absorption and in vivo anti-influenza activity of Stachyflin and its derivatives. Pharm Res. 1999;16:1041-6
66. Vichkanova SA, Oifa AI, Goriunova LV. [Antiviral properties of gossypol in experimental influenza pneumonia]. Antibiotiki. 1970;15:1071-3
67. Krylov VF. [Treatment of patients with influenza]. Ter Arkh. 1975;47:49-55
68. Yang J, Zhang F, Li J, Chen G, Wu S, Ouyang W. et al. Synthesis and antiviral activities of novel gossypol derivatives. Bioorg Med Chem Lett. 2012;22:1415-20
69. Yang J, Chen G, Li LL, Pan W, Zhang F, Yang J. et al. Synthesis and anti-H5N1 activity of chiral gossypol derivatives and its analogs implicated by a viral entry blocking mechanism. Bioorg Med Chem Lett. 2013;23:2619-23
70. Savov VM, Galabov AS, Tantcheva LP, Mileva MM, Pavlova EL, Stoeva ES. et al. Effects of rutin and quercetin on monooxygenase activities in experimental influenza virus infection. Exp Toxicol Pathol. 2006;58:59-64
71. Esanu V, Prahoveanu E, Crisan I, Cioca A. The effect of an aqueous propolis extract, of rutin and of a rutin-quercetin mixture on experimental influenza virus infection in mice. Virologie. 1981;32:213-5
72. Davis JM, Murphy EA, McClellan JL, Carmichael MD, Gangemi JD. Quercetin reduces susceptibility to influenza infection following stressful exercise. Am J Physiol Regul Integr Comp Physiol. 2008;295:R505-9
73. Choi HJ, Song JH, Park KS, Kwon DH. Inhibitory effects of quercetin 3-rhamnoside on influenza A virus replication. Eur J Pharm Sci. 2009;37:329-33
74. Kumar P, Khanna M, Srivastava V, Tyagi YK, Raj HG, Ravi K. Effect of quercetin supplementation on lung antioxidants after experimental influenza virus infection. Exp Lung Res. 2005;31:449-59
75. Chang TT, Sun MF, Chen HY, Tsai FJ, Fisher M, Lin JG. et al. Screening from the world's largest TCM database against H1N1 virus. J Biomol Struct Dyn. 2011;28:773-86
76. Nakayama M, Suzuki K, Toda M, Okubo S, Hara Y, Shimamura T. Inhibition of the infectivity of influenza virus by tea polyphenols. Antiviral Res. 1993;21:289-99
77. Zu M, Yang F, Zhou W, Liu A, Du G, Zheng L. In vitro anti-influenza virus and anti-inflammatory activities of theaflavin derivatives. Antiviral Res. 2012;94:217-24
78. Dang Z, Jung K, Zhu L, Xie H, Lee KH, Chen CH. et al. Phenolic diterpenoid derivatives as anti-influenza a virus agents. ACS Med Chem Lett. 2015;6:355-8
79. Yu M, Si L, Wang Y, Wu Y, Yu F, Jiao P. et al. Discovery of pentacyclic triterpenoids as potential entry inhibitors of influenza viruses. J Med Chem. 2014;57:10058-71
80. Chen X, Si L, Liu D, Proksch P, Zhang L, Zhou D. et al. Neoechinulin B and its analogues as potential entry inhibitors of influenza viruses, targeting viral hemagglutinin. Eur J Med Chem. 2015;93:182-95
81. Rossignol JF, Keeffe EB. Thiazolides: a new class of drugs for the treatment of chronic hepatitis B and C. Future Microbiol. 2008;3:539-45
82. Rossignol JF. Thiazolides: a new class of antiviral drugs. Expert Opin Drug Metab Toxicol. 2009;5:667-74
83. Rossignol JF. Nitazoxanide: a first-in-class broad-spectrum antiviral agent. Antiviral Res. 2014;110:94-103
84. Belardo G, Cenciarelli O, La Frazia S, Rossignol JF, Santoro MG. Synergistic effect of nitazoxanide with neuraminidase inhibitors against influenza A viruses in vitro. Antimicrob Agents Chemother. 2015;59:1061-9
85. Haffizulla J, Hartman A, Hoppers M, Resnick H, Samudrala S, Ginocchio C. et al. Effect of nitazoxanide in adults and adolescents with acute uncomplicated influenza: a double-blind, randomised, placebo-controlled, phase 2b/3 trial. Lancet Infect Dis. 2014;14:609-18
86. Ashton LV, Callan RL, Rao S, Landolt GA. In Vitro Susceptibility of Canine Influenza A (H3N8) Virus to Nitazoxanide and Tizoxanide. Vet Med Int. 2010. 2010
87. Colman PM, Ward CW. Structure and diversity of influenza virus neuraminidase. Curr Top Microbiol Immunol. 1985;114:177-255
88. Palese P, Tobita K, Ueda M, Compans RW. Characterization of temperature sensitive influenza virus mutants defective in neuraminidase. Virology. 1974;61:397-410
89. Liu C, Eichelberger MC, Compans RW, Air GM. Influenza type A virus neuraminidase does not play a role in viral entry, replication, assembly, or budding. J Virol. 1995;69:1099-106
90. Alymova IV, Taylor G, Portner A. Neuraminidase inhibitors as antiviral agents. Curr Drug Targets Infect Disord. 2005;5:401-9
91. Fladerer P, Wenisch C. [Neuraminidase inhibitors in therapy of influenza]. Acta Med Austriaca. 2000;27:133-40
92. Gubareva LV, Kaiser L, Hayden FG. Influenza virus neuraminidase inhibitors. Lancet. 2000;355:827-35
93. Palese P, Schulman JL, Bodo G, Meindl P. Inhibition of influenza and parainfluenza virus replication in tissue culture by 2-deoxy-2,3-dehydro-N-trifluoroacetylneuraminic acid (FANA). Virology. 1974;59:490-8
94. Ison MG. Clinical use of approved influenza antivirals: therapy and prophylaxis. Influenza Other Respir Viruses. 2013;7(Suppl 1):7-13
95. Meindl P, Bodo G, Lindner J, Palese P. [Influence of 2-deoxy-2,3-dehydro-N-acetylneuraminic acid on Myxovirus-neuraminidases and the replication of influenza- and Newcastle disease virus]. Z Naturforsch B. 1971;26:792-7
96. Ikeda K, Sano K, Ito M, Saito M, Hidari K, Suzuki T. et al. Synthesis of 2-deoxy-2,3-didehydro-N-acetylneuraminic acid analogues modified at the C-4 and C-9 positions and their behaviour towards sialidase from influenza virus and pig liver membrane. Carbohydr Res. 2001;330:31-41
97. Meindl P, Bodo G, Palese P, Schulman J, Tuppy H. Inhibition of neuraminidase activity by derivatives of 2-deoxy-2,3-dehydro-N-acetylneuraminic acid. Virology. 1974;58:457-63
98. Holzer CT, von Itzstein M, Jin B, Pegg MS, Stewart WP, Wu WY. Inhibition of sialidases from viral, bacterial and mammalian sources by analogues of 2-deoxy-2,3-didehydro-N-acetylneuraminic acid modified at the C-4 position. Glycoconj J. 1993;10:40-4
99. Varghese JN, Epa VC, Colman PM. Three-dimensional structure of the complex of 4-guanidino-Neu5Ac2en and influenza virus neuraminidase. Protein Sci. 1995;4:1081-7
100. Kim CU, Lew W, Williams MA, Liu H, Zhang L, Swaminathan S. et al. Influenza neuraminidase inhibitors possessing a novel hydrophobic interaction in the enzyme active site: design, synthesis, and structural analysis of carbocyclic sialic acid analogues with potent anti-influenza activity. J Am Chem Soc. 1997;119:681-90
101. Kohno S, Yen MY, Cheong HJ, Hirotsu N, Ishida T, Kadota J. et al. Phase III randomized, double-blind study comparing single-dose intravenous peramivir with oral oseltamivir in patients with seasonal influenza virus infection. Antimicrob Agents Chemother. 2011;55:5267-76
102. Yamashita M, Tomozawa T, Kakuta M, Tokumitsu A, Nasu H, Kubo S. CS-8958, a prodrug of the new neuraminidase inhibitor R-125489, shows long-acting anti-influenza virus activity. Antimicrob Agents Chemother. 2009;53:186-92
103. Hayden F. Developing new antiviral agents for influenza treatment: what does the future hold? Clin Infect Dis. 2009;48(Suppl 1):S3-13
104. Barroso L, Treanor J, Gubareva L, Hayden FG. Efficacy and tolerability of the oral neuraminidase inhibitor peramivir in experimental human influenza: randomized, controlled trials for prophylaxis and treatment. Antivir Ther. 2005;10:901-10
105. Watanabe A, Chang SC, Kim MJ, Chu DW, Ohashi Y. Long-acting neuraminidase inhibitor laninamivir octanoate versus oseltamivir for treatment of influenza: A double-blind, randomized, noninferiority clinical trial. Clin Infect Dis. 2010;51:1167-75
106. Sugaya N, Ohashi Y. Long-acting neuraminidase inhibitor laninamivir octanoate (CS-8958) versus oseltamivir as treatment for children with influenza virus infection. Antimicrob Agents Chemother. 2010;54:2575-82
107. Ikematsu H, Kawai N. Laninamivir octanoate: a new long-acting neuraminidase inhibitor for the treatment of influenza. Expert Rev Anti Infect Ther. 2011;9:851-7
108. Hanessian S, Wang J, Montgomery D, Stoll V, Stewart KD, Kati W. et al. Design, synthesis, and neuraminidase inhibitory activity of GS-4071 analogues that utilize a novel hydrophobic paradigm. Bioorg Med Chem Lett. 2002;12:3425-9
109. Bhatt B, Bohm R, Kerry PS, Dyason JC, Russell RJ, Thomson RJ. et al. Exploring the interactions of unsaturated glucuronides with influenza virus sialidase. J Med Chem. 2012;55:8963-8
110. Mohan S, McAtamney S, Haselhorst T, von Itzstein M, Pinto BM. Carbocycles related to oseltamivir as influenza virus group-1-specific neuraminidase inhibitors. Binding to N1 enzymes in the context of virus-like particles. J Med Chem. 2010;53:7377-91
111. Russell RJ, Haire LF, Stevens DJ, Collins PJ, Lin YP, Blackburn GM. et al. The structure of H5N1 avian influenza neuraminidase suggests new opportunities for drug design. Nature. 2006;443:45-9
112. Kerry PS, Mohan S, Russell RJ, Bance N, Niikura M, Pinto BM. Structural basis for a class of nanomolar influenza A neuraminidase inhibitors. Sci Rep. 2013;3:2871
113. Mohan S, Kerry PS, Bance N, Niikura M, Pinto BM. Serendipitous discovery of a potent influenza virus a neuraminidase inhibitor. Angew Chem Int Ed Engl. 2014;53:1076-80
114. Schade D, Kotthaus J, Riebling L, Kotthaus J, Muller-Fielitz H, Raasch W. et al. Development of novel potent orally bioavailable oseltamivir derivatives active against resistant influenza A. J Med Chem. 2014;57:759-69
115. Cheng TJ, Weinheimer S, Tarbet EB, Jan JT, Cheng YS, Shie JJ. et al. Development of oseltamivir phosphonate congeners as anti-influenza agents. J Med Chem. 2012;55:8657-70
116. Kim JH, Resende R, Wennekes T, Chen HM, Bance N, Buchini S. et al. Mechanism-based covalent neuraminidase inhibitors with broad-spectrum influenza antiviral activity. Science. 2013;340:71-5
117. McKimm-Breschkin JL, Colman PM, Jin B, Krippner GY, McDonald M, Reece PA. et al. Tethered neuraminidase inhibitors that bind an influenza virus: a first step towards a diagnostic method for influenza. Angew Chem Int Ed Engl. 2003;42:3118-21
118. Watson KG, Cameron R, Fenton RJ, Gower D, Hamilton S, Jin B. et al. Highly potent and long-acting trimeric and tetrameric inhibitors of influenza virus neuraminidase. Bioorg Med Chem Lett. 2004;14:1589-92
119. Macdonald SJ, Watson KG, Cameron R, Chalmers DK, Demaine DA, Fenton RJ. et al. Potent and long-acting dimeric inhibitors of influenza virus neuraminidase are effective at a once-weekly dosing regimen. Antimicrob Agents Chemother. 2004;48:4542-9
120. Macdonald SJ, Cameron R, Demaine DA, Fenton RJ, Foster G, Gower D. et al. Dimeric zanamivir conjugates with various linking groups are potent, long-lasting inhibitors of influenza neuraminidase including H5N1 avian influenza. J Med Chem. 2005;48:2964-71
121. Tarbet EB, Hamilton S, Vollmer AH, Luttick A, Ng WC, Pryor M. et al. A zanamivir dimer with prophylactic and enhanced therapeutic activity against influenza viruses. J Antimicrob Chemother. 2014;69:2164-74
122. Atigadda VR, Brouillette WJ, Duarte F, Babu YS, Bantia S, Chand P. et al. Hydrophobic benzoic acids as inhibitors of influenza neuraminidase. Bioorg Med Chem. 1999;7:2487-97
123. Brouillette WJ, Bajpai SN, Ali SM, Velu SE, Atigadda VR, Lommer BS. et al. Pyrrolidinobenzoic acid inhibitors of influenza virus neuraminidase: modifications of essential pyrrolidinone ring substituents. Bioorg Med Chem. 2003;11:2739-49
124. Chand P, Kotian PL, Morris PE, Bantia S, Walsh DA, Babu YS. Synthesis and inhibitory activity of benzoic acid and pyridine derivatives on influenza neuraminidase. Bioorg Med Chem. 2005;13:2665-78
125. Zhang J, Wang Q, Fang H, Xu W, Liu A, Du G. Design, synthesis, inhibitory activity, and SAR studies of hydrophobic p-aminosalicylic acid derivatives as neuraminidase inhibitors. Bioorg Med Chem. 2008;16:3839-47
126. Wang GT, Chen Y, Wang S, Gentles R, Sowin T, Kati W. et al. Design, Synthesis, and Structural Analysis of Influenza Neuraminidase Inhibitors Containing Pyrrolidine Cores. Journal of Medicinal Chemistry. 2001;44:1192-201
127. Maring CJ, Stoll VS, Zhao C, Sun M, Krueger AC, Stewart KD. et al. Structure-based characterization and optimization of novel hydrophobic binding interactions in a series of pyrrolidine influenza neuraminidase inhibitors. J Med Chem. 2005;48:3980-90
128. Krueger AC, Xu Y, Kati WM, Kempf DJ, Maring CJ, McDaniel KF. et al. Synthesis of potent pyrrolidine influenza neuraminidase inhibitors. Bioorg Med Chem Lett. 2008;18:1692-5
129. Zhang J, Wang Q, Fang H, Xu W, Liu A, Du G. Design, synthesis, inhibitory activity, and SAR studies of pyrrolidine derivatives as neuraminidase inhibitors. Bioorg Med Chem. 2007;15:2749-58
130. Miki K, Nagai T, Suzuki K, Tsujimura R, Koyama K, Kinoshita K. et al. Anti-influenza virus activity of biflavonoids. Bioorg Med Chem Lett. 2007;17:772-5
131. Ryu YB, Curtis-Long MJ, Kim JH, Jeong SH, Yang MS, Lee KW. et al. Pterocarpans and flavanones from Sophora flavescens displaying potent neuraminidase inhibition. Bioorg Med Chem Lett. 2008;18:6046-9
132. Li W, Liang H, Yin T, Wang B, Zhao YY. [Main flavonoids from Sophora flavescenes]. Yao Xue Xue Bao. 2008;43:833-7
133. Ryu YB, Curtis-Long MJ, Lee JW, Ryu HW, Kim JY, Lee WS. et al. Structural characteristics of flavanones and flavones from Cudrania tricuspidata for neuraminidase inhibition. Bioorg Med Chem Lett. 2009;19:4912-5
134. Lee CL, Chiang LC, Cheng LH, Liaw CC, Abd El-Razek MH, Chang FR. et al. Influenza A (H(1)N(1)) Antiviral and Cytotoxic Agents from Ferula assa-foetida. J Nat Prod. 2009;72:1568-72
135. Nagai T, Suzuki Y, Tomimori T, Yamada H. Antiviral activity of plant flavonoid, 5,7,4'-trihydroxy-8-methoxyflavone, from the roots of Scutellaria baicalensis against influenza A (H3N2) and B viruses. Biol Pharm Bull. 1995;18:295-9
136. Nagai T, Miyaichi Y, Tomimori T, Suzuki Y, Yamada H. In vivo anti-influenza virus activity of plant flavonoids possessing inhibitory activity for influenza virus sialidase. Antiviral Res. 1992;19:207-17
137. Ivachtchenko AV, Ivanenkov YA, Mitkin OD, Yamanushkin PM, Bichko VV, Shevkun NA. et al. Novel oral anti-influenza drug candidate AV5080. J Antimicrob Chemother. 2014;69:1892-902
138. Mooney CA, Johnson SA, t Hart P, Quarles van Ufford L, de Haan CA, Moret EE. et al. Oseltamivir analogues bearing N-substituted guanidines as potent neuraminidase inhibitors. J Med Chem. 2014;57:3154-60
139. Bauman JD, Patel D, Baker SF, Vijayan RS, Xiang A, Parhi AK. et al. Crystallographic fragment screening and structure-based optimization yields a new class of influenza endonuclease inhibitors. ACS Chem Biol. 2013;8:2501-8
140. Plotch SJ, Bouloy M, Ulmanen I, Krug RM. A unique cap(m7GpppXm)-dependent influenza virion endonuclease cleaves capped RNAs to generate the primers that initiate viral RNA transcription. Cell. 1981;23:847-58
141. Ruigrok RW, Crepin T, Hart DJ, Cusack S. Towards an atomic resolution understanding of the influenza virus replication machinery. Curr Opin Struct Biol. 2010;20:104-13
142. Olson AC, Rosenblum E, Kuchta RD. Regulation of influenza RNA polymerase activity and the switch between replication and transcription by the concentrations of the vRNA 5' end, the cap source, and the polymerase. Biochemistry. 2010;49:10208-15
143. Kerry PS, Willsher N, Fodor E. A cluster of conserved basic amino acids near the C-terminus of the PB1 subunit of the influenza virus RNA polymerase is involved in the regulation of viral transcription. Virology. 2008;373:202-10
144. Boivin S, Cusack S, Ruigrok RW, Hart DJ. Influenza A virus polymerase: structural insights into replication and host adaptation mechanisms. J Biol Chem. 2010;285:28411-7
145. Nakazawa M, Kadowaki SE, Watanabe I, Kadowaki Y, Takei M, Fukuda H. PA subunit of RNA polymerase as a promising target for anti-influenza virus agents. Antiviral Res. 2008;78:194-201
146. Naesens L, Stevaert A, Vanderlinden E. Antiviral therapies on the horizon for influenza. Curr Opin Pharmacol. 2016;30:106-15
147. Stevaert A, Naesens L. The Influenza Virus Polymerase Complex: An Update on Its Structure, Functions, and Significance for Antiviral Drug Design. Med Res Rev. 2016;36:1127-73
148. Su CY, Cheng TJ, Lin MI, Wang SY, Huang WI, Lin-Chu SY. et al. High-throughput identification of compounds targeting influenza RNA-dependent RNA polymerase activity. Proc Natl Acad Sci U S A. 2010;107:19151-6
149. Muratore G, Goracci L, Mercorelli B, Foeglein A, Digard P, Cruciani G. et al. Small molecule inhibitors of influenza A and B viruses that act by disrupting subunit interactions of the viral polymerase. Proc Natl Acad Sci U S A. 2012;109:6247-52
150. Massari S, Nannetti G, Goracci L, Sancineto L, Muratore G, Sabatini S. et al. Structural investigation of cycloheptathiophene-3-carboxamide derivatives targeting influenza virus polymerase assembly. J Med Chem. 2013;56:10118-31
151. Pagano M, Castagnolo D, Bernardini M, Fallacara AL, Laurenzana I, Deodato D. et al. The fight against the influenza A virus H1N1: synthesis, molecular modeling, and biological evaluation of benzofurazan derivatives as viral RNA polymerase inhibitors. ChemMedChem. 2014;9:129-50
152. Massari S, Nannetti G, Desantis J, Muratore G, Sabatini S, Manfroni G. et al. A Broad Anti-influenza Hybrid Small Molecule That Potently Disrupts the Interaction of Polymerase Acidic Protein-Basic Protein 1 (PA-PB1) Subunits. J Med Chem. 2015;58:3830-42
153. Shaw ML, Klumpp K. Successes and challenges in the antiviral field. Curr Opin Virol. 2013;3:483-6
154. Ulmanen I, Broni BA, Krug RM. Role of two of the influenza virus core P proteins in recognizing cap 1 structures (m7GpppNm) on RNAs and in initiating viral RNA transcription. Proc Natl Acad Sci U S A. 1981;78:7355-9
155. Blaas D, Patzelt E, Kuechler E. Identification of the cap binding protein of influenza virus. Nucleic Acids Res. 1982;10:4803-12
156. Guilligay D, Tarendeau F, Resa-Infante P, Coloma R, Crepin T, Sehr P. et al. The structural basis for cap binding by influenza virus polymerase subunit PB2. Nat Struct Mol Biol. 2008;15:500-6
157. Boyd MJ, Bandarage UK, Bennett H, Byrn RR, Davies I, Gu W. et al. Isosteric replacements of the carboxylic acid of drug candidate VX-787: Effect of charge on antiviral potency and kinase activity of azaindole-based influenza PB2 inhibitors. Bioorg Med Chem Lett. 2015;25:1990-4
158. Byrn RA, Jones SM, Bennett HB, Bral C, Clark MP, Jacobs MD. et al. Preclinical activity of VX-787, a first-in-class, orally bioavailable inhibitor of the influenza virus polymerase PB2 subunit. Antimicrob Agents Chemother. 2015;59:1569-82
159. Clark MP, Ledeboer MW, Davies I, Byrn RA, Jones SM, Perola E. et al. Discovery of a novel, first-in-class, orally bioavailable azaindole inhibitor (VX-787) of influenza PB2. J Med Chem. 2014;57:6668-78
160. Roch FF, Hinterkorner G, Menke J, Tang GQ, Cusack S, Butzendobler B. et al. An RNA Hybridization Assay for Screening Influenza A Virus Polymerase Inhibitors Using the Entire Ribonucleoprotein Complex. Assay Drug Dev Technol. 2015;13:488-506
161. Blair W, Cox C. Current Landscape of Antiviral Drug Discovery. F1000Research. 2016:5
162. Kuzuhara T, Iwai Y, Takahashi H, Hatakeyama D, Echigo N. Green tea catechins inhibit the endonuclease activity of influenza A virus RNA polymerase. PLoS currents. 2009;1:RRN1052
163. Iwai Y, Murakami K, Gomi Y, Hashimoto T, Asakawa Y, Okuno Y. et al. Anti-influenza activity of marchantins, macrocyclic bisbibenzyls contained in liverworts. PLoS One. 2011;6:e19825
164. Mori S, Miyake S, Kobe T, Nakaya T, Fuller SD, Kato N. et al. Enhanced anti-influenza A virus activity of (-)-epigallocatechin-3-O-gallate fatty acid monoester derivatives: effect of alkyl chain length. Bioorg Med Chem Lett. 2008;18:4249-52
165. Cianci C, Chung TDY, Meanwell N, Putz H, Hagen M, Colonno RJ. et al. Identification of N-hydroxamic acid and N-hydroxy-imide compounds that inhibit the influenza virus polymerase. Antivir Chem Chemother. 1996;7:353-60
166. Tomassini JE, Davies ME, Hastings JC, Lingham R, Mojena M, Raghoobar SL. et al. A novel antiviral agent which inhibits the endonuclease of influenza viruses. Antimicrob Agents Chemother. 1996;40:1189-93
167. Singh SB, Tomassini JE. Synthesis of natural flutimide and analogous fully substituted pyrazine-2,6-diones, endonuclease inhibitors of influenza virus. J Org Chem. 2001;66:5504-16
168. Parkes KE, Ermert P, Fassler J, Ives J, Martin JA, Merrett JH. et al. Use of a pharmacophore model to discover a new class of influenza endonuclease inhibitors. J Med Chem. 2003;46:1153-64
169. Stevaert A, Dallocchio R, Dessi A, Pala N, Rogolino D, Sechi M. et al. Mutational analysis of the binding pockets of the diketo acid inhibitor L-742,001 in the influenza virus PA endonuclease. J Virol. 2013;87:10524-38
170. Song MS, Kumar G, Shadrick WR, Zhou W, Jeevan T, Li Z. et al. Identification and characterization of influenza variants resistant to a viral endonuclease inhibitor. Proc Natl Acad Sci U S A. 2016;113:3669-74
171. Yuan S, Chu H, Singh K, Zhao H, Zhang K, Kao RY. et al. A novel small-molecule inhibitor of influenza A virus acts by suppressing PA endonuclease activity of the viral polymerase. Sci Rep. 2016;6:22880
172. Iwai Y, Takahashi H, Hatakeyama D, Motoshima K, Ishikawa M, Sugita K. et al. Anti-influenza activity of phenethylphenylphthalimide analogs derived from thalidomide. Bioorg Med Chem. 2010;18:5379-90
173. Shul'diakov AA, Liapina EP, Kuznetsov VI. [Current principles in the chemoprophylaxis of acute respiratory viral infections]. Ter Arkh. 2013;85:27-33
174. DuBois RM, Slavish PJ, Baughman BM, Yun MK, Bao J, Webby RJ. et al. Structural and biochemical basis for development of influenza virus inhibitors targeting the PA endonuclease. PLoS Pathog. 2012;8:e1002830
175. Shoji M, Takahashi E, Hatakeyama D, Iwai Y, Morita Y, Shirayama R. et al. Anti-influenza activity of c60 fullerene derivatives. PLoS One. 2013;8:e66337
176. Sagong HY, Parhi A, Bauman JD, Patel D, Vijayan RS, Das K. et al. 3-Hydroxyquinolin-2(1H)-ones As Inhibitors of Influenza A Endonuclease. ACS Med Chem Lett. 2013;4:547-50
177. Parhi AK, Xiang A, Bauman JD, Patel D, Vijayan RS, Das K. et al. Phenyl substituted 3-hydroxypyridin-2(1H)-ones: inhibitors of influenza A endonuclease. Bioorg Med Chem. 2013;21:6435-46
178. Sagong HY, Bauman JD, Patel D, Das K, Arnold E, LaVoie EJ. Phenyl substituted 4-hydroxypyridazin-3(2H)-ones and 5-hydroxypyrimidin-4(3H)-ones: inhibitors of influenza A endonuclease. J Med Chem. 2014;57:8086-98
179. Fudo S, Yamamoto N, Nukaga M, Odagiri T, Tashiro M, Hoshino T. Two Distinctive Binding Modes of Endonuclease Inhibitors to the N-Terminal Region of Influenza Virus Polymerase Acidic Subunit. Biochemistry. 2016;55:2646-60
180. Fudo S, Yamamoto N, Nukaga M, Odagiri T, Tashiro M, Neya S. et al. Structural and computational study on inhibitory compounds for endonuclease activity of influenza virus polymerase. Bioorg Med Chem. 2015;23:5466-75
181. Chen E, Swift RV, Alderson N, Feher VA, Feng GS, Amaro RE. Computation-guided discovery of influenza endonuclease inhibitors. ACS Med Chem Lett. 2014;5:61-4
182. Carcelli M, Rogolino D, Bacchi A, Rispoli G, Fisicaro E, Compari C. et al. Metal-chelating 2-hydroxyphenyl amide pharmacophore for inhibition of influenza virus endonuclease. Mol Pharm. 2014;11:304-16
183. Rogolino D, Bacchi A, De Luca L, Rispoli G, Sechi M, Stevaert A. et al. Investigation of the salicylaldehyde thiosemicarbazone scaffold for inhibition of influenza virus PA endonuclease. J Biol Inorg Chem. 2015;20:1109-21
184. Stevaert A, Nurra S, Pala N, Carcelli M, Rogolino D, Shepard C. et al. An integrated biological approach to guide the development of metal-chelating inhibitors of influenza virus PA endonuclease. Mol Pharmacol. 2015;87:323-37
185. Pala N, Stevaert A, Dallocchio R, Dessi A, Rogolino D, Carcelli M. et al. Virtual Screening and Biological Validation of Novel Influenza Virus PA Endonuclease Inhibitors. ACS Med Chem Lett. 2015;6:866-71
186. Baughman BM, Jake Slavish P, DuBois RM, Boyd VA, White SW, Webb TR. Identification of influenza endonuclease inhibitors using a novel fluorescence polarization assay. ACS Chem Biol. 2012;7:526-34
187. Sidwell RW, Khare GP, Allen LB, Huffman JG, Witkowski JT, Simon LN. et al. In vitro and in vivo effect of 1-beta-D-ribofuranosyl-1,2,4-triazole-3-carboxamide (ribavirin) on types 1 and 3 parainfulenza virus infections. Chemotherapy. 1975;21:205-20
188. Furuta Y, Takahashi K, Fukuda Y, Kuno M, Kamiyama T, Kozaki K. et al. In vitro and in vivo activities of anti-influenza virus compound T-705. Antimicrob Agents Chemother. 2002;46:977-81
189. Takahashi K, Furuta Y, Fukuda Y, Kuno M, Kamiyama T, Kozaki K. et al. In vitro and in vivo activities of T-705 and oseltamivir against influenza virus. Antivir Chem Chemother. 2003;14:235-41
190. Allen LB, Boswell KH, Khwaja TA, Meyer RB Jr, Sidwell RW, Witkowski JT. et al. Synthesis and antiviral acticity of some phosphates of the broad-spectrum antiviral nucleoside, 1-beta-D-ribofuranosyl-1,2,4-triazole-3-carboxamide (ribavirin). J Med Chem. 1978;21:742-6
191. Goswami BB, Borek E, Sharma OK, Fujitaki J, Smith RA. The broad spectrum antiviral agent ribavirin inhibits capping of mRNA. Biochem Biophys Res Commun. 1979;89:830-6
192. Chill DW, Johnston PB. Inhibition of measles virus replication and enhancement of cellular DNA synthesis in vero cells by ribavirin, an antiviral and antineoplastic drug. Zhonghua Minguo wei sheng wu xue za zhi = Chinese journal of microbiology. 1975;8:67-72
193. Oxford JS. Inhibition of the replication of influenza A and B viruses by a nucleoside analogue (ribavirin). J Gen Virol. 1975;28:409-14
194. Pauly MD, Lauring AS. Effective lethal mutagenesis of influenza virus by three nucleoside analogs. J Virol. 2015;89:3584-97
195. Naesens L, Guddat LW, Keough DT, van Kuilenburg AB, Meijer J, Vande Voorde J. et al. Role of human hypoxanthine guanine phosphoribosyltransferase in activation of the antiviral agent T-705 (favipiravir). Mol Pharmacol. 2013;84:615-29
196. Wang G, Wan J, Hu Y, Wu X, Prhavc M, Dyatkina N. et al. Synthesis and Anti-Influenza Activity of Pyridine, Pyridazine, and Pyrimidine C-Nucleosides as Favipiravir (T-705) Analogues. J Med Chem. 2016;59:4611-24
197. Gao J, Luo X, Li Y, Gao R, Chen H, Ji D. Synthesis and biological evaluation of 2-oxo-pyrazine-3-carboxamide-yl nucleoside analogues and their epimers as inhibitors of influenza A viruses. Chem Biol Drug Des. 2015;85:245-52
198. Furuta Y, Takahashi K, Shiraki K, Sakamoto K, Smee DF, Barnard DL. et al. T-705 (favipiravir) and related compounds: Novel broad-spectrum inhibitors of RNA viral infections. Antiviral Res. 2009;82:95-102
199. Sleeman K, Mishin VP, Deyde VM, Furuta Y, Klimov AI, Gubareva LV. In vitro antiviral activity of favipiravir (T-705) against drug-resistant influenza and 2009 A(H1N1) viruses. Antimicrob Agents Chemother. 2010;54:2517-24
200. Tisdale M, Appleyard G, Tuttle JV, Nelson DJ, Nusinoff-Lehrman S, Al Nakib W. et al. Inhibition of influenza A and B viruses by 2_-deoxy-2_-fluororibosides. Antivir Chem Chemother. 1993;4:281-7
201. Vedula MS, Jennepalli S, Aryasomayajula R, Rondla SR, Musku MR, Kura RR. et al. Novel nucleosides as potent influenza viral inhibitors. Bioorg Med Chem. 2010;18:6329-39
202. Abdel-Magid AF. Influenza RNA-Dependent RNA Polymerase (RdRp) Inhibitors: Potential New Therapy for Influenza Treatment. ACS Med Chem Lett. 2013;4:1133-4
203. Lin C, Sun C, Liu X, Zhou Y, Hussain M, Wan J. et al. Design, synthesis, and in vitro biological evaluation of novel 6-methyl-7-substituted-7-deaza purine nucleoside analogs as anti-influenza A agents. Antiviral Res. 2016;129:13-20
204. Kumaki Y, Day CW, Smee DF, Morrey JD, Barnard DL. In vitro and in vivo efficacy of fluorodeoxycytidine analogs against highly pathogenic avian influenza H5N1, seasonal, and pandemic H1N1 virus infections. Antiviral Res. 2011;92:329-40
205. Mena I, Jambrina E, Albo C, Perales B, Ortin J, Arrese M. et al. Mutational analysis of influenza A virus nucleoprotein: identification of mutations that affect RNA replication. J Virol. 1999;73:1186-94
206. Ozawa M, Fujii K, Muramoto Y, Yamada S, Yamayoshi S, Takada A. et al. Contributions of two nuclear localization signals of influenza A virus nucleoprotein to viral replication. J Virol. 2007;81:30-41
207. Portela A, Digard P. The influenza virus nucleoprotein: a multifunctional RNA-binding protein pivotal to virus replication. J Gen Virol. 2002;83:723-34
208. Ye Q, Krug RM, Tao YJ. The mechanism by which influenza A virus nucleoprotein forms oligomers and binds RNA. Nature. 2006;444:1078-82
209. Gerritz SW, Cianci C, Kim S, Pearce BC, Deminie C, Discotto L. et al. Inhibition of influenza virus replication via small molecules that induce the formation of higher-order nucleoprotein oligomers. Proc Natl Acad Sci U S A. 2011;108:15366-71
210. Kao RY, Yang D, Lau LS, Tsui WH, Hu L, Dai J. et al. Identification of influenza A nucleoprotein as an antiviral target. Nat Biotechnol. 2010;28:600-5
211. Cheng H, Wan J, Lin MI, Liu Y, Lu X, Liu J. et al. Design, synthesis, and in vitro biological evaluation of 1H-1,2,3-triazole-4-carboxamide derivatives as new anti-influenza A agents targeting virus nucleoprotein. J Med Chem. 2012;55:2144-53
212. Pons M. Effect of actinomycin D on the replication of influenza virus and influenza virus RNA. Virology. 1967;33:150-4
213. Pons MW. The inhibition of influenza virus RNA synthesis by actinomycin D and cycloheximide. Virology. 1973;51:120-8
214. Vogel U, Scholtissek C. Inhibition of the intracellular transport of influenza viral RNA by actinomycin D. Arch Virol. 1995;140:1715-23
215. Lejal N, Tarus B, Bouguyon E, Chenavas S, Bertho N, Delmas B. et al. Structure-based discovery of the novel antiviral properties of naproxen against the nucleoprotein of influenza A virus. Antimicrob Agents Chemother. 2013;57:2231-42
216. Semenova NP, Prokudina EN, Livov DK, Nebol'sin VE. [Effect of the antiviral drug Ingaviruin on intracellular transformations and import into the nucleus of influenza A virus nucleocapsid protein]. Vopr Virusol. 2010;55:17-20
217. Loginova S, Borisevich SV, Shkliaeva OM, Maksimov VA, Bondarev VP, Nebol'sin VE. [Prophylactic and therapeutic efficacies of Ingavirin, a novel Russian chemotherapeutic, with respect to influenza pathogen A (H5N1)]. Antibiot Khimioter. 2010;55:10-2
218. Zarubaev VV, Beliaevskaia SV, Sirotkin AK, Anfimov PM, Nebol'sin VE, Kiselev OI. et al. [In vitro and in vivo effects of ingavirin on the ultrastructure and infectivity of influenza virus]. Vopr Virusol. 2011;56:21-5
219. Galegov GA, Andronova VL, Nebol'sin VE. [Antiviral effect of Ingavirin against seasonal influenza virus A/H1N1 in MDCK cell culture]. Antibiot Khimioter. 2009;54:19-22
220. Shishkina LN, Nebol'sin VE, Kabanov AS, Skarnovich MO, Mazurkova NA, Sergeev AA. et al. [In vitro and in vivo efficacy of Ingavirin against strains of pandemic influenza virus A(H1N1/09)v]. Zh Mikrobiol Epidemiol Immunobiol. 2011:93-6
221. Shishkina LN, Nebol'sin VE, Skarnovich MO, Kabanov AS, Sergeev AA, Erdyneeva UB. et al. [In vivo efficacy of Ingavirin against pandemic A (H1N1/09)v influenza virus]. Antibiot Khimioter. 2010;55:32-5
222. Stouffer AL, Acharya R, Salom D, Levine AS, Di Costanzo L, Soto CS. et al. Structural basis for the function and inhibition of an influenza virus proton channel. Nature. 2008;451:596-9
223. Tang Y, Zaitseva F, Lamb RA, Pinto LH. The gate of the influenza virus M2 proton channel is formed by a single tryptophan residue. J Biol Chem. 2002;277:39880-6
224. Holsinger LJ, Nichani D, Pinto LH, Lamb RA. Influenza A virus M2 ion channel protein: a structure-function analysis. J Virol. 1994;68:1551-63
225. Pielak RM, Chou JJ. Influenza M2 proton channels. Biochim Biophys Acta. 2011;1808:522-9
226. Schnell JR, Chou JJ. Structure and mechanism of the M2 proton channel of influenza A virus. Nature. 2008;451:591-5
227. Du QS, Huang RB, Wang SQ, Chou KC. Designing inhibitors of M2 proton channel against H1N1 swine influenza virus. PLoS One. 2010;5:e9388
228. Hong M, DeGrado WF. Structural basis for proton conduction and inhibition by the influenza M2 protein. Protein Sci. 2012;21:1620-33
229. Acharya R, Carnevale V, Fiorin G, Levine BG, Polishchuk AL, Balannik V. et al. Structure and mechanism of proton transport through the transmembrane tetrameric M2 protein bundle of the influenza A virus. Proc Natl Acad Sci U S A. 2010;107:15075-80
230. Tataridis D, Fytas G, Kolocouris A, Fytas C, Kolocouris N, Foscolos GB. et al. Influence of an additional 2-amino substituent of the 1-aminoethyl pharmacophore group on the potency of rimantadine against influenza virus A. Bioorg Med Chem Lett. 2007;17:692-6
231. Wang J, Wu Y, Ma C, Fiorin G, Wang J, Pinto LH. et al. Structure and inhibition of the drug-resistant S31N mutant of the M2 ion channel of influenza A virus. Proc Natl Acad Sci U S A. 2013;110:1315-20
232. Wu Y, Canturk B, Jo H, Ma C, Gianti E, Klein ML. et al. Flipping in the pore: discovery of dual inhibitors that bind in different orientations to the wild-type versus the amantadine-resistant S31N mutant of the influenza A virus M2 proton channel. J Am Chem Soc. 2014;136:17987-95
233. Rey-Carrizo M, Torres E, Ma C, Barniol-Xicota M, Wang J, Wu Y. et al. 3-Azatetracyclo[5.2.1.1(5,8).0(1,5)]undecane derivatives: from wild-type inhibitors of the M2 ion channel of influenza A virus to derivatives with potent activity against the V27A mutant. J Med Chem. 2013;56:9265-74
234. Wang J, Ma C, Wang J, Jo H, Canturk B, Fiorin G. et al. Discovery of novel dual inhibitors of the wild-type and the most prevalent drug-resistant mutant, S31N, of the M2 proton channel from influenza A virus. J Med Chem. 2013;56:2804-12
235. Tanner JA, Zheng BJ, Zhou J, Watt RM, Jiang JQ, Wong KL. et al. The adamantane-derived bananins are potent inhibitors of the helicase activities and replication of SARS coronavirus. Chem Biol. 2005;12:303-11
236. Moorthy NS, Poongavanam V, Pratheepa V. Viral M2 ion channel protein: a promising target for anti-influenza drug discovery. Mini Rev Med Chem. 2014;14:819-30
237. Bachrach U, Don S. Inactivation of influenza and Newcastle disease viruses by oxidized spermine. Isr J Med Sci. 1970;6:435-7
238. Bachrach U. Antiviral activity of oxidized polyamines. Amino Acids. 2007;33:267-72
239. Lin TI, Heider H, Schroeder C. Different modes of inhibition by adamantane amine derivatives and natural polyamines of the functionally reconstituted influenza virus M2 proton channel protein. J Gen Virol. 1997;78( Pt 4):767-74
240. Even-Or O, Samira S, Rochlin E, Balasingam S, Mann AJ, Lambkin-Williams R. et al. Immunogenicity, protective efficacy and mechanism of novel CCS adjuvanted influenza vaccine. Vaccine. 2010;28:6527-41
241. Even-Or O, Joseph A, Itskovitz-Cooper N, Samira S, Rochlin E, Eliyahu H. et al. A new intranasal influenza vaccine based on a novel polycationic lipid-ceramide carbamoyl-spermine (CCS). II. Studies in mice and ferrets and mechanism of adjuvanticity. Vaccine. 2011;29:2474-86
242. Zhao X, Jie Y, Rosenberg MR, Wan J, Zeng S, Cui W. et al. Design and synthesis of pinanamine derivatives as anti-influenza A M2 ion channel inhibitors. Antiviral Res. 2012;96:91-9
243. Galabov AS, Velichkova E, Karparov A, Sidzhakova D, Danchev D, Chakova N. Antiviral activity of tetrahydro-2(1H)-pyrimidinones and related compounds. Arzneimittelforschung. 1984;34:9-14
244. Karparov A, Galabov AS, Sidzhakova D, Danchev D. Antiviral effect of 1-morpholinomethyl-tetrahydro-2(1H)-pyrimidinone (DD-13) in experimental alphaviral infections in white mice. Arzneimittelforschung. 1985;35:1269-75
245. Tverdislov VA, Kharitonenkov IG, Sukhanov SV, Galabov AS. [Effect of a new antiviral compound 1-morpholinomethyltetrahydro-2(1H)-pyrimidinone on the interaction of influenza virus proteins with flat lipid membranes]. Vopr Virusol. 1988;33:278-81
246. Galabov AS, Khristova ML, Uzunov S, Wassilewa L, Bucher DJ, Kharitonenkov IG. Alteration in the antigenic structure of M1 protein of influenza A virus mutant resistant to a new antiviral compound mopyridone. Acta Virol. 1994;38:5-10
247. Tantcheva LP, Rangelova DS. Toxicity and enzyme-inducing effect of the antiviral compound mopyridone in mice. Arzneimittelforschung. 1996;46:931-3
248. Leneva IA, Fediakina IT, Gus'kova TA, Glushkov RG. [Sensitivity of various influenza virus strains to arbidol. Influence of arbidol combination with different antiviral drugs on reproduction of influenza virus A]. Ter Arkh. 2005;77:84-8
249. Boriskin YS, Leneva IA, Pecheur EI, Polyak SJ. Arbidol: a broad-spectrum antiviral compound that blocks viral fusion. Curr Med Chem. 2008;15:997-1005
250. Boriskin YS, Pecheur EI, Polyak SJ. Arbidol: a broad-spectrum antiviral that inhibits acute and chronic HCV infection. Virol J. 2006;3:56
251. Wang MZ, Cai BQ, Li LY, Lin JT, Su N, Yu HX. et al. [Efficacy and safety of arbidol in treatment of naturally acquired influenza]. Zhongguo Yi Xue Ke Xue Yuan Xue Bao. 2004;26:289-93
252. Leneva IA, Russell RJ, Boriskin YS, Hay AJ. Characteristics of arbidol-resistant mutants of influenza virus: Implications for the mechanism of anti-influenza action of arbidol. Antiviral Res. 2009;81:132-40
253. Horimoto T, Nakayama K, Smeekens SP, Kawaoka Y. Proprotein-processing endoproteases PC6 and furin both activate hemagglutinin of virulent avian influenza viruses. J Virol. 1994;68:6074-8
254. Stieneke-Grober A, Vey M, Angliker H, Shaw E, Thomas G, Roberts C. et al. Influenza virus hemagglutinin with multibasic cleavage site is activated by furin, a subtilisin-like endoprotease. EMBO J. 1992;11:2407-14
255. Tashiro M, Klenk HD, Rott R. Inhibitory effect of a protease inhibitor, leupeptin, on the development of influenza pneumonia, mediated by concomitant bacteria. J Gen Virol. 1987;68( Pt 7):2039-41
256. Puzis LE, Lozitsky VP. Action of epsilon-aminocaproic acid on the proteolysis system during experimental influenza in mice. Acta Virol. 1988;32:515-21
257. Lee MG, Kim KH, Park KY, Kim JS. Evaluation of anti-influenza effects of camostat in mice infected with non-adapted human influenza viruses. Arch Virol. 1996;141:1979-89
258. Zhirnov OP, Klenk HD, Wright PF. Aprotinin and similar protease inhibitors as drugs against influenza. Antiviral Res. 2011;92:27-36
259. Gong J, Fang H, Li M, Liu Y, Yang K, Liu Y. et al. Potential targets and their relevant inhibitors in anti-influenza fields. Curr Med Chem. 2009;16:3716-39
260. Ott S, Wunderli-Allenspach H. Effect of the virostatic Norakin (triperiden) on influenza virus activities. Antiviral Res. 1994;24:37-42
261. Presber HW, Schroeder C, Hegenscheid B, Heider H, Reefschlager J, Rosenthal HA. Antiviral activity of Norakin (triperiden) and related anticholinergic antiparkinsonism drugs. Acta Virol. 1984;28:501-7
262. Heider H, Markushin S, Schroeder C, Ghendon Y. The influence of Norakin on the reproduction of influenza A and B viruses. Arch Virol. 1985;86:283-90
263. Sugiyama K, Obayashi E, Kawaguchi A, Suzuki Y, Tame JR, Nagata K. et al. Structural insight into the essential PB1-PB2 subunit contact of the influenza virus RNA polymerase. EMBO J. 2009;28:1803-11
264. Ananenko AA, Malinovskaia VV, Klembovskii AI, Politova LN, Pertseva NG. [Lipid peroxidation in experimental viral infection and the effect on it of interferons and alpha-tocopherol]. Vopr Med Khim. 1988;34:86-9
265. Poryvaev VD, Zykova NA. [The effect of conditions of storage and composition of viral material on lipid peroxidation of the viral envelope and inactivation of the influenza virus]. Vopr Virusol. 1995;40:62-5
266. Gardner EM, Bernstein ED, Popoff KA, Abrutyn E, Gross P, Murasko DM. Immune response to influenza vaccine in healthy elderly: lack of association with plasma beta-carotene, retinol, alpha-tocopherol, or zinc. Mech Ageing Dev. 2000;117:29-45
267. Chase G, Wunderlich K, Reuther P, Schwemmle M. Identification of influenza virus inhibitors which disrupt of viral polymerase protein-protein interactions. Methods. 2011;55:188-91
268. Zhang XH, Zhong X, Zhou YM, Du HM, Wang T. Effect of RRR-alpha-tocopherol succinate on the growth and immunity in broilers. Poult Sci. 2009;88:959-66
269. Walker WT, Faust SN. Monovalent inactivated split-virion AS03-adjuvanted pandemic influenza A (H1N1) vaccine. Expert review of vaccines. 2010;9:1385-98
270. Morel S, Didierlaurent A, Bourguignon P, Delhaye S, Baras B, Jacob V. et al. Adjuvant System AS03 containing alpha-tocopherol modulates innate immune response and leads to improved adaptive immunity. Vaccine. 2011;29:2461-73
271. Garcon N, Vaughn DW, Didierlaurent AM. Development and evaluation of AS03, an Adjuvant System containing alpha-tocopherol and squalene in an oil-in-water emulsion. Expert review of vaccines. 2012;11:349-66
272. Galabov AS, Mileva M, Simeonova L, Gegova G. Combination activity of neuraminidase inhibitor oseltamivir and alpha-tocopherol in influenza virus A (H3N2) infection in mice. Antivir Chem Chemother. 2015;24:83-91
273. Akaike T, Noguchi Y, Ijiri S, Setoguchi K, Suga M, Zheng YM. et al. Pathogenesis of influenza virus-induced pneumonia: involvement of both nitric oxide and oxygen radicals. Proc Natl Acad Sci U S A. 1996;93:2448-53
274. Imai Y, Kuba K, Neely GG, Yaghubian-Malhami R, Perkmann T, van Loo G. et al. Identification of oxidative stress and Toll-like receptor 4 signaling as a key pathway of acute lung injury. Cell. 2008;133:235-49
275. Suliman HB, Ryan LK, Bishop L, Folz RJ. Prevention of influenza-induced lung injury in mice overexpressing extracellular superoxide dismutase. Am J Physiol Lung Cell Mol Physiol. 2001;280:L69-78
276. Oda T, Akaike T, Hamamoto T, Suzuki F, Hirano T, Maeda H. Oxygen radicals in influenza-induced pathogenesis and treatment with pyran polymer-conjugated SOD. Science. 1989;244:974-6
277. Kash JC, Xiao Y, Davis AS, Walters KA, Chertow DS, Easterbrook JD. et al. Treatment with the reactive oxygen species scavenger EUK-207 reduces lung damage and increases survival during 1918 influenza virus infection in mice. Free Radic Biol Med. 2014;67:235-47
278. Ye S, Lowther S, Stambas J. Inhibition of reactive oxygen species production ameliorates inflammation induced by influenza A viruses via upregulation of SOCS1 and SOCS3. J Virol. 2015;89:2672-83
279. Selemidis S, Seow HJ, Broughton BR, Vinh A, Bozinovski S, Sobey CG. et al. Nox1 oxidase suppresses influenza a virus-induced lung inflammation and oxidative stress. PLoS One. 2013;8:e60792
280. Vlahos R, Selemidis S. NADPH oxidases as novel pharmacologic targets against influenza A virus infection. Mol Pharmacol. 2014;86:747-59
281. Planz O. Development of cellular signaling pathway inhibitors as new antivirals against influenza. Antiviral Res. 2013;98:457-68
282. Krug RM, Yuan W, Noah DL, Latham AG. Intracellular warfare between human influenza viruses and human cells: the roles of the viral NS1 protein. Virology. 2003;309:181-9
283. Nemeroff ME, Barabino SM, Li Y, Keller W, Krug RM. Influenza virus NS1 protein interacts with the cellular 30 kDa subunit of CPSF and inhibits 3'end formation of cellular pre-mRNAs. Mol Cell. 1998;1:991-1000
284. Engelhardt OG, Fodor E. Functional association between viral and cellular transcription during influenza virus infection. Rev Med Virol. 2006;16:329-45
285. Noah DL, Twu KY, Krug RM. Cellular antiviral responses against influenza A virus are countered at the posttranscriptional level by the viral NS1A protein via its binding to a cellular protein required for the 3' end processing of cellular pre-mRNAS. Virology. 2003;307:386-95
286. Kochs G, Garcia-Sastre A, Martinez-Sobrido L. Multiple anti-interferon actions of the influenza A virus NS1 protein. J Virol. 2007;81:7011-21
287. Twu KY, Noah DL, Rao P, Kuo RL, Krug RM. The CPSF30 binding site on the NS1A protein of influenza A virus is a potential antiviral target. J Virol. 2006;80:3957-65
288. Jablonski JJ, Basu D, Engel DA, Geysen HM. Design, synthesis, and evaluation of novel small molecule inhibitors of the influenza virus protein NS1. Bioorg Med Chem. 2012;20:487-97
289. Selvy PE, Lavieri RR, Lindsley CW, Brown HA. Phospholipase D: enzymology, functionality, and chemical modulation. Chem Rev. 2011;111:6064-119
290. O'Reilly MC, Oguin TH 3rd, Scott SA, Thomas PG, Locuson CW, Morrison RD. et al. Discovery of a highly selective PLD2 inhibitor (ML395): a new probe with improved physiochemical properties and broad-spectrum antiviral activity against influenza strains. ChemMedChem. 2014;9:2633-7
291. Boulo S, Akarsu H, Ruigrok RW, Baudin F. Nuclear traffic of influenza virus proteins and ribonucleoprotein complexes. Virus Res. 2007;124:12-21
292. Paterson D, Fodor E. Emerging roles for the influenza A virus nuclear export protein (NEP). PLoS Pathog. 2012;8:e1003019
293. Paragas J, Talon J, O'Neill RE, Anderson DK, Garcia-Sastre A, Palese P. Influenza B and C virus NEP (NS2) proteins possess nuclear export activities. J Virol. 2001;75:7375-83
294. Perwitasari O, Johnson S, Yan X, Howerth E, Shacham S, Landesman Y. et al. Verdinexor, a novel selective inhibitor of nuclear export, reduces influenza a virus replication in vitro and in vivo. J Virol. 2014;88:10228-43
295. Drago L, Nicola L, Ossola F, De Vecchi E. In vitro antiviral activity of resveratrol against respiratory viruses. J Chemother. 2008;20:393-4
296. Palamara AT, Nencioni L, Aquilano K, De Chiara G, Hernandez L, Cozzolino F. et al. Inhibition of influenza A virus replication by resveratrol. J Infect Dis. 2005;191:1719-29
297. Li C, Fang JS, Lian WW, Pang XC, Liu AL, Du GH. In vitro antiviral effects and 3D QSAR study of resveratrol derivatives as potent inhibitors of influenza H1N1 neuraminidase. Chem Biol Drug Des. 2015;85:427-38
298. Furuya A, Uozaki M, Yamasaki H, Arakawa T, Arita M, Koyama AH. Antiviral effects of ascorbic and dehydroascorbic acids in vitro. Int J Mol Med. 2008;22:541-5
299. Uozaki M, Ikeda K, Tsujimoto K, Nishide M, Yamasaki H, Khamsri B. et al. Antiviral effects of dehydroascorbic acid. Exp Ther Med. 2010;1:983-6
300. Khanna M, Saxena L, Rajput R, Kumar B, Prasad R. Gene silencing: a therapeutic approach to combat influenza virus infections. Future Microbiol. 2015;10:131-40
301. Fedson DS. Treating influenza with statins and other immunomodulatory agents. Antiviral Res. 2013;99:417-35
302. Mortensen EM, Nakashima B, Cornell J, Copeland LA, Pugh MJ, Anzueto A. et al. Population-based study of statins, angiotensin II receptor blockers, and angiotensin-converting enzyme inhibitors on pneumonia-related outcomes. Clin Infect Dis. 2012;55:1466-73
303. Rothberg MB, Bigelow C, Pekow PS, Lindenauer PK. Association between statins given in hospital and mortality in pneumonia patients. J Gen Intern Med. 2012;27:280-6
304. Vandermeer ML, Thomas AR, Kamimoto L, Reingold A, Gershman K, Meek J. et al. Association between use of statins and mortality among patients hospitalized with laboratory-confirmed influenza virus infections: a multistate study. J Infect Dis. 2012;205:13-9
305. Darwish I, Mubareka S, Liles WC. Immunomodulatory therapy for severe influenza. Expert Rev Anti Infect Ther. 2011;9:807-22
306. Eisen DP, Reid D, McBryde ES. Acetyl salicylic acid usage and mortality in critically ill patients with the systemic inflammatory response syndrome and sepsis. Crit Care Med. 2012;40:1761-7
307. Di Raimondo D, Tuttolomondo A, Butta C, Miceli S, Licata G, Pinto A. Effects of ACE-inhibitors and angiotensin receptor blockers on inflammation. Curr Pharm Des. 2012;18:4385-413
308. Wong AK, Howie J, Petrie JR, Lang CC. AMP-activated protein kinase pathway: a potential therapeutic target in cardiometabolic disease. Clin Sci (Lond). 2009;116:607-20
309. Paumelle R, Staels B. Cross-talk between statins and PPARalpha in cardiovascular diseases: clinical evidence and basic mechanisms. Trends Cardiovasc Med. 2008;18:73-8
310. Birnbaum Y, Ye Y, Lin Y, Freeberg SY, Nishi SP, Martinez JD. et al. Augmentation of myocardial production of 15-epi-lipoxin-a4 by pioglitazone and atorvastatin in the rat. Circulation. 2006;114:929-35
311. Yano M, Matsumura T, Senokuchi T, Ishii N, Murata Y, Taketa K. et al. Statins activate peroxisome proliferator-activated receptor gamma through extracellular signal-regulated kinase 1/2 and p38 mitogen-activated protein kinase-dependent cyclooxygenase-2 expression in macrophages. Circ Res. 2007;100:1442-51
312. Fedson DS. Confronting an influenza pandemic with inexpensive generic agents: can it be done? Lancet Infect Dis. 2008;8:571-6
313. Fedson DS. Confronting the next influenza pandemic with anti-inflammatory and immunomodulatory agents: why they are needed and how they might work. Influenza Other Respir Viruses. 2009;3:129-42
314. Caldeira D, Alarcao J, Vaz-Carneiro A, Costa J. Risk of pneumonia associated with use of angiotensin converting enzyme inhibitors and angiotensin receptor blockers: systematic review and meta-analysis. BMJ. 2012;345:e4260
315. Govorkova EA, McCullers JA. Therapeutics against influenza. Curr Top Microbiol Immunol. 2013;370:273-300
316. Ilyushina NA, Bovin NV, Webster RG, Govorkova EA. Combination chemotherapy, a potential strategy for reducing the emergence of drug-resistant influenza A variants. Antiviral Res. 2006;70:121-31
317. Govorkova EA, Webster RG. Combination chemotherapy for influenza. Viruses. 2010;2:1510-29
318. Ilyushina NA, Hay A, Yilmaz N, Boon AC, Webster RG, Govorkova EA. Oseltamivir-ribavirin combination therapy for highly pathogenic H5N1 influenza virus infection in mice. Antimicrob Agents Chemother. 2008;52:3889-97
319. Ma Y, Zhang W, Zhao Z, Li M, Liu J, Wang Y. Combination of ribavirin and reduning protects mice against severe pneumonia induced by H1N1 influenza a virus. J Tradit Chin Med. 2016;36:181-6
320. Ilyushina NA, Hoffmann E, Salomon R, Webster RG, Govorkova EA. Amantadine-oseltamivir combination therapy for H5N1 influenza virus infection in mice. Antivir Ther. 2007;12:363-70
321. Nguyen JT, Hoopes JD, Le MH, Smee DF, Patick AK, Faix DJ. et al. Triple combination of amantadine, ribavirin, and oseltamivir is highly active and synergistic against drug resistant influenza virus strains in vitro. PLoS One. 2010;5:e9332
322. Gasparini R, Amicizia D, Lai PL, Bragazzi NL, Panatto D. Compounds with anti-influenza activity: present and future of strategies for the optimal treatment and management of influenza. Part II: Future compounds against influenza virus. J Prev Med Hyg. 2014;55:109-29
Author contact
![]() Corresponding author: Dr. Zhiyun Jia is to be contacted at Department of Nuclear Medicine, West China Hospital, Sichuan University, No. 37 Guo Xue Xiang, Chengdu, Sichuan (610041), PR China. E-mail address: jzygoodedu.cn (Z. Jia). Tel.: +86-18980605783; fax: + 86 28 85423503.
Corresponding author: Dr. Zhiyun Jia is to be contacted at Department of Nuclear Medicine, West China Hospital, Sichuan University, No. 37 Guo Xue Xiang, Chengdu, Sichuan (610041), PR China. E-mail address: jzygoodedu.cn (Z. Jia). Tel.: +86-18980605783; fax: + 86 28 85423503.
 Global reach, higher impact
Global reach, higher impact