13.3
Impact Factor
Theranostics 2016; 6(13):2352-2366. doi:10.7150/thno.15433 This issue Cite
Research Paper
ROS-Responsive Mitochondria-Targeting Blended Nanoparticles: Chemo- and Photodynamic Synergistic Therapy for Lung Cancer with On-Demand Drug Release upon Irradiation with a Single Light Source
1. Institute of Nano Biomedicine and Engineering, Key Laboratory for Thin Film and Microfabrication Technology of the Ministry of Education, Shanghai, Engineering Center for Intelligent Diagnosis and Treatment Instrument, Department of Instrument Science & Engineering, School of Electronic Information and Electrical Engineering, Shanghai Jiao Tong University, Shanghai 200240, P. R. China;
2. School of Biomedical Engineering, Shanghai Jiao Tong University, Shanghai 200240, P. R. China;
3. Department of Oncology, Tongren Hospital, Shanghai Jiao Tong University School of Medicine, Shanghai 200336, P. R. China;
4. National Center for Translational Medicine, Collaborative Innovational Center for System Biology, Shanghai Jiao Tong University, Shanghai 200240, P. R. China.
Abstract
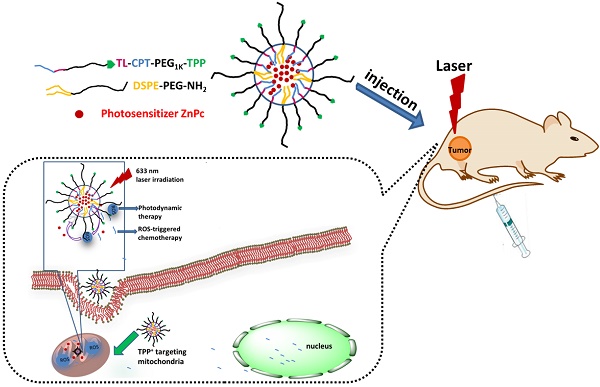
Mitochondria in cancer cells maintain a more negative membrane potential than normal cells. Mitochondria are the primary source of cellular reactive oxygen species (ROS), which are necessary for photodynamic therapy. Thus, the strategy of targeting mitochondria can maximize the photodynamic therapeutic efficiency for cancer. Here we report, for the first time, synthesis of a new mitochondria-targeting drug delivery system, ZnPc/CPT-TPPNPs. To synthesize this novel compound, polyethylene glycol was functionalized with thioketal linker-modified camptothecin (TL-CPT) and triphenylphosphonium to form the block copolymer, TL-CPT-PEG1K-TPP. The ZnPc/CPT-TPPNPs was constructed for delivery of the photosensitizer Zinc phthalocyanine (ZnPc) by blending the block copolymer TL-CPT-PEG1K-TPP with 1, 2-distearoyl-sn-glycero-3-phosphoethanolamine-N-[methoxy (polyethylene glycol)] (DSPE-PEG).Triphenylphosphine can accumulate selectively several hundred-fold within mitochondria. The thioketal linker is ROS-responsive and CPT can be released upon ROS cleavage. We also show that the ZnPc loaded in ZnPc/CPT-TPPNPs absorbed the 633 nm laser to produce ROS, which could be utilized both in photodynamic therapy and to cleave the thioketal linker thereby releasing camptothecin for chemotherapy. Thus, the mitochondria-targeting nanoparticles could elevate photodynamic therapeutic efficacy. Our results showed that surface modification of the nanoparticles with triphenylphosphine cations facilitated efficient subcellular delivery of the photosensitizer to mitochondria. The nanoparticles had a good ROS-responsive effect to release CPT, which could transfer to the nucleus and interfere with DNA replication as a topoisomeraseⅠinhibitor. Thus, the blended nanoparticles provide a new promising approach as a mitochondria-targeting ROS-activated chemo- and photodynamic therapy with a single light source for lung cancer.
Keywords: mitochondria-targeting nanoparticles, ROS-responsive, Zinc phthalocyanine, photodynamic therapy, camptothecin.
 Global reach, higher impact
Global reach, higher impact