13.3
Impact Factor
Theranostics 2016; 6(9):1415-1424. doi:10.7150/thno.15250 This issue Cite
Research Paper
Colorimetric TMPRSS2-ERG Gene Fusion Detection in Prostate Cancer Urinary Samples via Recombinase Polymerase Amplification
1. Centre for Personalized Nanomedicine, Australian Institute for Bioengineering and Nanotechnology (AIBN), The University of Queensland, QLD 4072, Australia;
2. School of Chemistry and Molecular Biosciences, The University of Queensland, QLD 4072, Australia.
Abstract
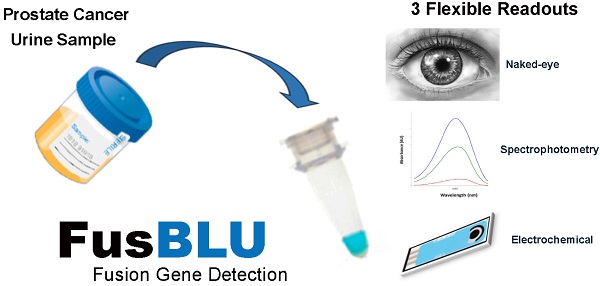
TMPRSS2 (Exon 1)-ERG (Exon 4) is the most frequent gene fusion event in prostate cancer (PC), and is highly PC-specific unlike the current serum prostate specific antigen (PSA) biomarker. However, TMPRSS2-ERG levels are currently measured with quantitative reverse-transcription PCR (RT-qPCR) which is time-consuming and requires costly equipment, thus limiting its use in clinical diagnostics. Herein, we report a novel rapid, cost-efficient and minimal-equipment assay named “FusBLU” for detecting TMPRSS2-ERG gene fusions from urine. TMPRSS2-ERG mRNA was amplified by isothermal reverse transcription-recombinase polymerase amplification (RT-RPA), magnetically-isolated, and detected through horseradish peroxidase (HRP)-catalyzed colorimetric reaction. FusBLU was specific for TMPRSS2-ERG mRNA with a low visual detection limit of 105 copies. We also demonstrated assay readout versatility on 3 potentially useful platforms. The colorimetric readout was detectable by naked eye for a quick yes/no evaluation of gene fusion presence. On the other hand, a more quantitative TMPRSS2-ERG detection was achievable by absorbance/electrochemical measurements. FusBLU was successfully applied to 12 urinary samples and results were validated by gold-standard RT-qPCR. We also showed that sediment RNA was likely the main source of TMPRSS2-ERG mRNA in urinary samples. We believe that our assay is a potential clinical screening tool for PC and could also have wide applications for other disease-related fusion genes.
Keywords: TMPRSS2-ERG, FusBLU
 Global reach, higher impact
Global reach, higher impact