13.3
Impact Factor
Theranostics 2016; 6(8):1085-1095. doi:10.7150/thno.13448 This issue Cite
Research Paper
Design of Internalizing PSMA-specific Glu-ureido-based Radiotherapeuticals
1. AffiliationDepartment for Nuclear Medicine, Heidelberg University Hospital, Im Neuenheimer Feld 400, 69120 Heidelberg, Germany
2. Division of Radiopharmaceutical Chemistry, German Cancer Research Center (DKFZ), Heidelberg, Germany
3. German Cancer Consortium (DKTK), Heidelberg, Germany.
Abstract
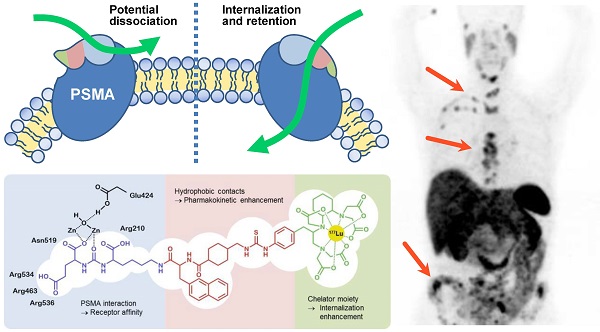
Despite the progress in diagnosis and treatment, prostate cancer (PCa) is one of the main causes for cancer-associated deaths among men. Recently, prostate-specific membrane antigen (PSMA) binding tracers have revolutionized the molecular imaging of this disease. The translation of these tracers into therapeutic applications is challenging because of high PSMA-associated kidney uptake. While both the tumor uptake and the uptake in the kidneys are PSMA-specific, the kidneys show a more rapid clearance than tumor lesions. Consequently, the potential of endoradiotherapeutic drugs targeting PSMA is highly dependent on a sustained retention in the tumor - ideally achieved by predominant internalization of the respective tracer. Previously, we were able to show that the pharmacokinetics of the tracers containing the Glu-urea-based binding motif can be further enhanced with a specifically designed linker. Here, we evaluate an eventual influence of the chelator moiety on the pharmacokinetics, including the tumor internalization. A series of tracers modified by different chelators were synthesized using solid phase chemistry. The conjugates were radiolabeled to evaluate the influence on the receptor binding affinity, the ligand-induced internalization and the biodistribution behavior. Competitive binding and internalization assays were performed on PSMA positive LNCaP cells and the biodistribution of the most promising compound was evaluated by positron emission tomography (PET) in LNCaP-tumor-bearing mice. Interestingly, conjugation of the different chelators did not cause significant differences: all compounds showed nanomolar binding affinities with only minor differences. PET imaging of the 68Ga-labeled CHX-A''-DTPA conjugate revealed that the chelator moiety does not impair the specificity of tumor uptake when compared to the gold standard PSMA-617. However, strong differences of the internalization ratios caused by the chelator moiety were observed: differences in internalization between 15% and 65% were observed, with the CHX-A''-DTPA conjugate displaying the highest internalization ratio. A first-in-man PET/CT study proved the high tumor uptake of this 68Ga-labeled PSMA-targeting compound. These data indicate that hydrophobic entities at the chelator mediate the internalization efficacy. Based on its specific tumor uptake in combination with its very high internalization ratio, the clinical performance of the chelator-conjugated Glu-urea-based PSMA inhibitors will be further elucidated.
Keywords: prostate-specific membrane antigen, PSMA, tumor targeting, prostate cancer, chelators, theranostics, positron emission tomography
 Global reach, higher impact
Global reach, higher impact