Impact Factor
Theranostics 2016; 6(6):887-895. doi:10.7150/thno.14742 This issue Cite
Research Paper
Conformationally Strained trans-Cyclooctene (sTCO) Enables the Rapid Construction of 18F-PET Probes via Tetrazine Ligation
1. Department of Radiology and Biomedical Research Imaging Center, University of North Carolina at Chapel Hill, Chapel Hill, North Carolina, 27599, United States.
2. Brown Laboratories, Department of Chemistry and Biochemistry, University of Delaware, Newark, Delaware 19716, United States.
3. Institute of Applied Synthetic Chemistry, TU Wien, Vienna, 1060, Austria.
Abstract
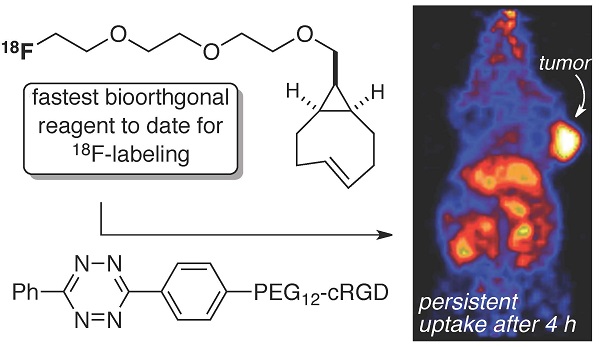
The bioorthogonal reaction between tetrazines and trans-cyclooctenes is a method for the rapid construction of F-18 probes for PET imaging. Described here is a second generation 18F-labeling system based on a conformationally strained trans-cyclooctene (sTCO)—a dienophile that is approximately 2 orders of magnitude more reactive than conventional TCO dienophiles. Starting from a readily prepared tosylate precursor, an 18F labeled sTCO derivative (18F-sTCO) could be synthesized in 29.3 +/- 5.1% isolated yield and with high specific activity. Tetrazine ligation was carried out with a cyclic RGD-conjugate of a diphenyl-s-tetrazine analogue (RGD-Tz) chosen from a diene class with an excellent combination of fast reactivity and stability both for the diene as well as the Diels-Alder adduct. For both the tetrazine and the sTCO, mini-PEG spacers were included to enhance solubility and improve the in vivo distribution profile of the resulting probe. Extremely fast reactivity (up to 2.86 x 105 M-1s-1 at 25 °C in water) has been observed in kinetic studies in the reaction of sTCO with diphenyl-s-tetrazine derivatives. A kinetic study on sTCO diastereomers in 55:45 MeOH:water showed that the syn-diastereomer displayed slightly faster reactivity than the anti-diastereomer. An 18F-sTCO conjugate with RGD-Tz demonstrated prominent and persistent tumor uptake in vivo with good tumor-to-background contrast. Unlike most radiolabeled RGD peptides, the tumor uptake of this PET agent increased from 5.3 +/- 0.2% ID/g at 1 h post injection (p.i.), to 8.9 +/- 0.5% ID/g at 4 h p.i., providing evidence for prolonged blood circulation. These findings suggest that tetrazine ligations employing 18F-sTCO should serve as a powerful and general platform for the rapid construction of peptide or protein derived PET agents.
Keywords: bioorthogonal reaction, strained trans-cyclooctene (sTCO), positron emission tomography (PET), 18F, RGD peptide.
 Global reach, higher impact
Global reach, higher impact