13.3
Impact Factor
Theranostics 2015; 5(4):399-417. doi:10.7150/thno.10351 This issue Cite
Research Paper
Targeted Antiangiogenesis Gene Therapy Using Targeted Cationic Microbubbles Conjugated with CD105 Antibody Compared with Untargeted Cationic and Neutral Microbubbles
1. Chongqing Key Laboratory of Ultrasound Molecular Imaging, Institute of Ultrasound Imaging, The Second Affiliated Hospital of Chongqing Medical University, Chongqing 400010, P. R. China.
2. Department of Ultrasound, The Children's Hospital of Chongqing Medical University, Chongqing 400014, P. R. China.
3. Department of Gastrointestinal & Anorectal Surgery, The Second Affiliated Hospital of Chongqing Medical University, Chongqing 400010, P. R. China.
4. Department of Endocrine & Breast Surgery, The First Affiliated Hospital of Chongqing Medical University, Chongqing 400016, P. R. China.
5. Department of Otolaryngology, The Third People's Hospital of Chongqing, Chongqing 400014, P. R. China.
6. Department of Ophthalmology, The Second Affiliated Hospital of Chongqing Medical University, Chongqing 400010, P. R. China.
7. Chongqing Jinxin Advertisement Company, Chongqing Daily News Group, Chongqing 400012, P. R. China.
Abstract
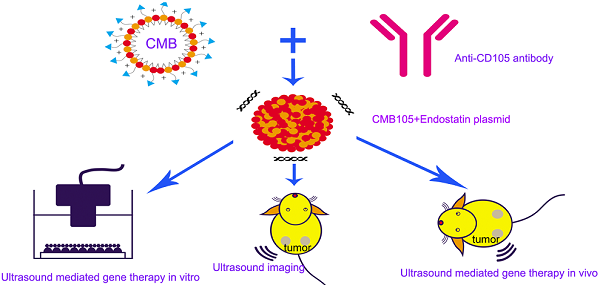
Objective This study aimed to develop targeted cationic microbubbles conjugated with a CD105 antibody (CMB105) for use in targeted vascular endothelial cell gene therapy and ultrasound imaging. We compared the results with untargeted cationic microbubbles (CMB) and neutral microbubbles (NMB).
Methods CMB105 were prepared and compared with untargeted CMB and NMB. First, the microbubbles were characterized in terms of size, zeta-potential, antibody binding ability and plasmid DNA loading capacity. A tumor model of subcutaneous breast cancer in nude mice was used for our experiments. The ability of different types of microbubbles to target HUVECs in vitro and tumor neovascularization in vivo was measured. The endostatin gene was selected for its outstanding antiangiogenesis effect. For in vitro experiments, the transfection efficiency and cell cycle were analyzed using flow cytometry, and the transcription and expression of endostatin were measured by qPCR and Western blotting, respectively. Vascular tube cavity formation and tumor cell invasion were used to evaluate the antiangiogenesis gene therapy efficiency in vitro. Tumors were exposed to ultrasound irradiation with different types of microbubbles, and the gene therapy effects were investigated by detecting apoptosis induction and changes in tumor volume.
Results CMB105 and CMB differed significantly from NMB in terms of zeta-potential, and the DNA loading capacities were 16.76±1.75 μg, 18.21±1.22 μg, and 0.48±0.04 μg per 5×108 microbubbles, respectively. The charge coupling of plasmid DNA to CMB105 was not affected by the presence of the CD105 antibody. Both CMB105 and CMB could target to HUVECs in vitro, whereas only CMB105 could target to tumor neovascularization in vivo. In in vitro experiments, the transfection efficiency of CMB105 was 24.7-fold higher than the transfection efficiency of NMB and 1.47-fold higher than the transfection efficiency of CMB (P<0.05). With ultrasound-targeted microbubble destruction (UTMD)-mediated gene therapy, the transcription and expression of endostatin were the highest in the CMB105 group (P<0.001); the antiangiogenesis effect and inhibition of tumor cells invasion was better with CMB105 than CMB or NMB in vitro (P<0.01). After gene therapy, the tumor volumes of CMB105 group were significantly smaller than that of CMB and NMB, and many tumor cells had begun apoptosis in the CMB105 group, which had the highest apoptosis index (P<0.001).
Conclusions As a contrast agent and plasmid carrier, CMB105 can be used not only for targeted ultrasound imaging but also for targeted gene therapy both in vitro and in vivo. The plasmid DNA binding ability of the CMB was not affected by conjugation of the CMB with the CD105 antibody, and because of its targeting ability, the gene transfection efficiency and therapeutic effect were better compared with the untargeted CMB and NMB. The advantages of targeted gene therapy with CMB105 in vivo were more prominent than with CMB or NMB because neither can target the endothelia in vivo.
Keywords: Ultrasound-mediated gene delivery (UMGD), Antiangiogenesis, Target, Cationic microbubbles
 Global reach, higher impact
Global reach, higher impact