Impact Factor
Theranostics 2014; 4(9):893-903. doi:10.7150/thno.9425 This issue Cite
Research Paper
An EGFR Targeted PET Imaging Probe for the Detection of Colonic Adenocarcinomas in the Setting of Colitis
1. Athinoula A. Martinos Center for Biomedical Imaging, Department of Radiology, Massachusetts General Hospital, Harvard Medical School, Boston, MA;
2. Department of Medicine/Division of Genetics, Brigham and Women's Hospital, Harvard Medical School, Boston, MA.
Abstract
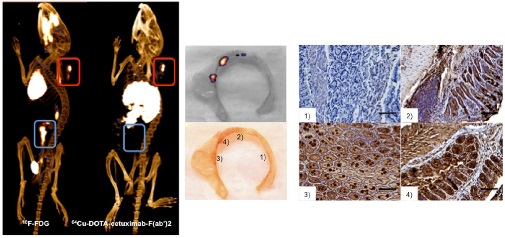
Colorectal cancer is a serious complication associated with inflammatory bowel disease, often indistinguishable by screening with conventional FDG PET probes. We have developed an alternative EGFR-targeted PET imaging probe that may be used to overcome this difficulty, and successfully assessed its utility for neoplastic lesion detection in preclinical models. Cetuximab F(ab′)2 fragments were enzymatically generated, purified, and DOTA-conjugated. Radiolabeling was performed with 67Ga for cell based studies and 64Cu for in vivo imaging. Competitive binding studies were performed on CT26 cells to assess affinity (KD) and receptors per cell (Bmax). In vivo imaging using the EGFR targeted PET probe and 18F FDG was performed on CT26 tumor bearing mice in both control and dextran sodium sulfate (DSS) induced colitis settings. Spontaneous adenomas in genetically engineered mouse (GEM) models of colon cancer were additionally imaged. The EGFR imaging agent was generated with high purity (> 98%), with a labeling efficiency of 60 ± 5% and ≥99% radiochemical purity. The KD was 6.6 ± 0.7 nM and the Bmax for CT26 cells was 3.3 ± 0.1 × 106 receptors/cell. Target to background ratios (TBR) for CT26 tumors compared to colonic uptake demonstrated high values for both 18F-FDG (3.95 ± 0.13) and the developed 64Cu-DOTA-cetuximab-F(ab′)2 probe (4.42 ± 0.11) in control mice. The TBR for the EGFR targeted probe remained high (3.78 ± 0.06) in the setting of colitis, while for 18F FDG, this was markedly reduced (1.54 ± 0.08). Assessment of the EGFR targeted probe in the GEM models demonstrated a correlation between radiotracer uptake in spontaneous colonic lesions and the EGFR staining level ex vivo. A clinically translatable PET imaging probe was successfully developed to assess EGFR. The imaging agent can detect colonic tumors with a high TBR for detection of in situ lesions in the setting of colitis, and opens the possibility for a new approach for screening high-risk patients.
Keywords: Positron emission tomography (PET) imaging, molecular imaging, EGFR, ulcerative colitis, colorectal cancer, mouse models.
 Global reach, higher impact
Global reach, higher impact