13.3
Impact Factor
Theranostics 2013; 3(9):618-632. doi:10.7150/thno.6810 This issue Cite
Research Paper
Improved PET Imaging of uPAR Expression Using new 64Cu-labeled Cross-Bridged Peptide Ligands: Comparative in vitro and in vivo Studies
1. The Danish-Chinese Center for Proteases and Cancer;
2. Finsen Laboratory, Rigshospitalet & BRIC, Copenhagen Biocenter, Denmark;
3. Cluster for Molecular Imaging, Faculty of Health Sciences, University of Copenhagen, Copenhagen, Denmark;
4. Department of Clinical Physiology, Nuclear Medicine and PET, Center for Diagnostic Investigations, Rigshospitalet, Copenhagen, Denmark;
5. Department of Chemistry, University of Copenhagen, Denmark;
6. Department of Biochemistry and Molecular Biology, University of Southern Denmark, Denmark.
Abstract
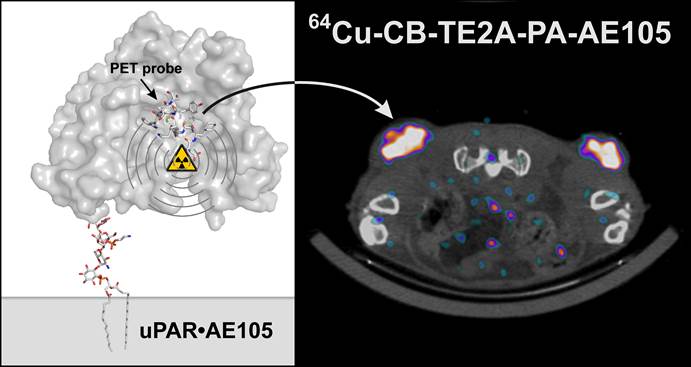
The correlation between uPAR expression, cancer cell invasion and metastases is now well-established and has prompted the development of a number of uPAR PET imaging agents, which could potentially identify cancer patients with invasive and metastatic lesions. In the present study, we synthesized and characterized two new cross-bridged 64Cu-labeled peptide conjugates for PET imaging of uPAR and performed a head-to-head comparison with the corresponding and more conventionally used DOTA conjugate. Based on in-source laser-induced reduction of chelated Cu(II) to Cu(I), we now demonstrate the following ranking with respect to the chemical inertness of their complexed Cu ions: DOTA-AE105 << CB-TE2A-AE105 < CB-TE2A-PA-AE105, which is correlated to their corresponding demetallation rate. No penalty in the uPAR receptor binding affinity of the targeting peptide was encountered by conjugation to either of the macrobicyclic chelators (IC50 ~ 5-10 nM) and high yields and radiochemical purities (>95%) were achieved in all cases by incubation at 95ºC. In vivo, they display identical tumor uptake after 1h, but differ significantly after 22 hrs, where the DOTA-AE105 uptake remains surprisingly high. Importantly, the more stable of the new uPAR PET tracers, 64Cu-CB-TE2A-PA-AE105, exhibits a significantly reduced liver uptake compared to 64Cu-DOTA-AE105 as well as 64Cu-CB-TE2A-AE105, (p<0.0001), emphasizing that our new in vitro stability measurements by mass spectrometry predicts in vivo stability in mice. Specificity of the best performing ligand, 64Cu-CB-TE2A-PA-AE105 was finally confirmed in vivo using a non-binding 64Cu-labeled peptide as control (64Cu-CB-TE2A-PA-AE105mut). This control PET-tracer revealed significantly reduced tumor uptake (p<0.0001), but identical hepatic uptake compared to its active counterpart (64Cu-CB-TE2A-PA-AE105) after 1h. In conclusion, our new approach using in-source laser-induced reduction of Cu(II)-chelated PET-ligands provides useful information, which are predictive for the tracer stability in vivo in mice. Furthermore, the increased stability of our new macrobicyclic 64Cu-CB-TE2A-PA-AE105 PET ligand is paralleled by an excellent imaging contrast during non-invasive PET scanning of uPAR expression in preclinical mouse cancer models. The translational promises displayed by this PET-tracer for future clinical cancer patient management remains, however, to be investigated.
Keywords: urokinase-type plasminogen activator receptor, CD87, positron emission tomography, biomarker, CB-TE2A, Ly-6, SPR, tumor targeting.
 Global reach, higher impact
Global reach, higher impact