13.3
Impact Factor
Theranostics 2012; 2(3):313-317. doi:10.7150/thno.4007 This issue Cite
Review
Clinical Indications of 11C-Choline PET/CT in Prostate Cancer Patients with Biochemical Relapse
1. Nuclear Medicine Department, San Raffaele Scientific Institute, Milan, Italy; Institute for Bioimaging and Molecular Physiology, National Research Council (IBFM-CNR), Milan, Italy;
2. Nuclear Medicine Unit, Azienda Ospedaliero-Universitaria di Bologna Policlinico S.Orsola-Malpighi, University of Bologna, Italy.
Received 2011-12-23; Accepted 2012-1-18; Published 2012-3-12
Abstract
Several studies investigated the potential role of imaging modalities in prostate cancer patients in case of biochemical recurrence. However, the role of molecular imaging has not been well established yet. Considering the results of the literature and of our own experience, we tried to summarize the potential applications of 11C-choline PET/CT in prostate cancer patients in case of biochemical relapse for the detection of lymph node and distant recurrence.
Keywords: prostate cancer, choline PET/CT, restaging.
Background
Monitoring Prostate Specific Antigen (PSA) serum level is the best way to follow Prostate Cancer (PC) patients after treatment and to detect early recurrence of the disease [1]. In case of biochemical relapse (PSA>0.2-0.4 ng/mL), Imaging should be able to detect the site of recurrence in order to establish a correct and tailored therapeutic strategy. It is crucial to distinguish between the presence of local failure and/or distant metastasis [2]. The Conventional Imaging (C.I.: Bone Scan; Computed Tomography or Magnetic Resonance) accuracy is limited in this field as reported by many authors [3, 4, 5, 6]. The site of distant recurrence, mainly lymph-nodes or bone lesions, is thus, rarely detected. In the clinical daily practice it results in an enormous number of negative or inconclusive investigations performed in this setting of patients. Choueiri et al [5] tried to identify, in 292 patients with biochemical failure, clinical parameters that were predictive of positive Imaging studies, that could be helpful in guiding clinicians in their requests. Authors found that age, imaging type Computed Tomography (CT), Magnetic Resonace (MR), Bone scan (BS), PSA and PSA doubling time (PSAdt) were significantly associated with positive imaging results. However, CT, MR and BS were not useful when PSA was lower than 5 ng/mL and PSAdt greater than 10 months. Summarizing, C.I. showed a sensitivity of only 11% (31/292) with a mean PSA value of 23 ng/mL in the group of positive patients. In a similar study published by Okotie et al [6], the authors addressed their attention to the influence of PSA and PSAdt on CT and BS sensitivity. Bone scan was positive in only 11% and CT scan in only 7% of the patients.
The low accuracy shown by C.I. has meant that the attention of several authors has been directed to other imaging techniques and molecular imaging in particular.
11C-Choline PET/CT
The study of tissue metabolism using PET/CT could play an important role in this context [7]. In the field of prostate cancer, many studies have been addressed to this aim in the early past since the first study published by Hara et al in 1998 [8] about the potential use of 11C-choline in prostate cancer patients. In this section we will try to highlight the potentials, but also the limitations shown by 11C-choline in this field.
The first large and prospective study was performed by Picchio et al [9] comparing 11C-choline PET with 18F-FDG PET results in 100 prostate cancer patients with a biochemical recurrence (mean PSA value 6.57 ng/mL). 11C-choline PET detected areas of abnormal uptake in 47% of patients, while 18F-FDG PET in 27% of patients. None of FDG positive was negative at 11C-choline. C.I. (BS and MR) were positive in 49 patients. The study stated the utility of 11C-choline PET/CT in the restaging of prostate cancer patients after relapse: in particular the authors affirmed that 11C-choline PET resulted to be superior to 18F-FDG PET and complementary to C.I., providing almost the same sensitivity as C.I. but by performing a single examination. It is worth to underline however that this study was performed with PET only scanners and not with PET/CT.
In the following years many studies focused their attention to the correlation between the detection rate of 11C-choline PET/CT and the serum PSA levels. Krause et al [10] evaluated with 11C-choline PET/CT sixty-three prostate cancer patients with biochemical relapse after primary treatment (mean PSA 5,9 ng/mL). They demonstrated a significant and strict correlation between 11C-choline PET/CT detection rate and PSA serum levels: the detection rate was 36% for a PSA value <1 ng/mL, 43% for a PSA value 1-<2 ng/mL, 62% for a PSA value 2-<3 ng/mL and 73% for a PSA value >or=3 ng/mL. The overall detection rate for PET was 59%.
The influence of other clinical and pathological variables also was evaluated in order to better select patients which should undergo 11C-choline PET/CT. Giovacchini et al [11] published the results of their investigations on 358 prostate cancer patients (mean PSA 3.7 ng/mL): they found that in multivariate analysis not only PSA levels, but also advanced pathological stage, previous biochemical failure and older age (> 65 y) were significantly associated with an increased risk of positive 11C-choline PET/CT findings.
In 2009, Castellucci et al [12] for the first time investigated the relationship between 11C-choline PET/CT detection rate and other PSA derivates in particular PSA kinetics (PSA velocity: PSA vel; PSA doubling time: PSAdt). Authors enrolled a total of 190 patients after radical prostatectomy (RP) who showed an increase in PSA (mean 4.2 ng/mL; median 2.1 ng/mL). The study showed an overall detection rate of 38.9%. PSA values, PSAdt and PSAvel values were the only parameters who showed a statistically significant difference between patients with PET-positive and PET-negative findings. Authors conclude that PSA kinetics should always be taken into consideration before performing a 11C-choline PET/CT in patients with biochemical failure because it is the most relevant positive predictive factor. Later on, another study by Giovacchini et al [13] confirmed these data. They evaluated the influence of PSAdt in 170 patients studied with 11C-choline PET/CT after RP who showed a biochemical failure (mean PSA 3.2 ng/mL). The study showed an overall detection rate of 44% and an overall accuracy of 88%. Multivariate logistic regression found that high PSA values and short PSAdt were the only significant predictors of positive 11C-choline PET/CT scan. Moreover, their results demonstrated that PSAdt can distinguish between patients with pathological 11C-choline uptake in the skeleton from patients with pathological 11C-choline uptake in the prostatic fossa. Since kinetics of PSA has been found to be the most important factor in predicting a positive 11C-choline PET/CT, Castellucci et al [14] have selected a population of 102 consecutive patients previously treated with radical prostatectomy (RP) and who presented only a mild increase of PSA levels <1.5 ng/mL. The aim of the study was to determine if 11C-choline PET/CT could be used also in patients with low PSA values but fast PSA kinetics, i.e. early after biochemical recurrence when the clinical usefulness of the investigation should be maximal. Overall, 11C-choline PET/CT showed positive findings in 29 of 102 patients (28%). What resulted very interesting was that the mean PSAdt in PET-positive patients was 4.34 months while in PET-negative patients it was 13.30 months (p = 0.0001 ). Moreover, only 2 out of 56 patients (3%) with slow kinetics (PSAdt <7.2 months) resulted positive at 11C-choline PET/CT versus 27 out of 46 patients (58%) with fast kinetics that resulted positive at 11C-choline PET/CT. Authors concluded that 11C-choline PET/CT could be suggested early during biochemical relapse in patients presenting with fast PSA kinetics. Fig.1.
68y.o. patient. Prostate Cancer T3aN1Mx. Biochemical relapse two years after radical prostatectomy. PSA = 1.3 ng/mL at the time of 11C-Choline PET/CT. PSAdt = 3 months. 11C-Choline PET /CT detected a single positive lymph-node in the left iliac chain. a) CT images, a small sub-centimetric LN is evident in the left iliac chain b) focal increased uptake of 11C-Choline in the left iliac chain c) fused images d) MIP.
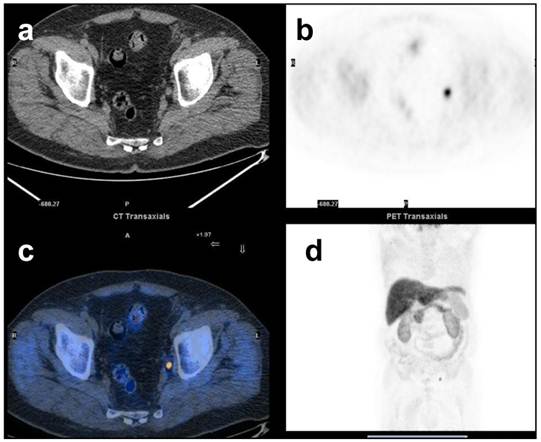
As remembered above one of the sites more frequently involved by metastatic lesions from prostate cancer is the skeleton. However only few studies have performed a direct comparison between the accuracy of 11C-choline PET/CT and the one of Bone Scan in the detection of bone lesions.
Fuccio et al [15] studied with 11C-choline PET/CT twenty-five patients who presented a single finding that was classified as equivocal or suspected for metastatic lesion at BS. Authors found that 11C-choline PET/CT detected unknown lesions in 11/25 (44%) patients. Very recently in a larger study, Picchio et al [16] studied 78 patients with biochemical progression (mean PSA 21.1 ng/ml) with 11C-choline PET/CT and BS. Authors found that equivocal findings occurred in only 1 of 78 (1%) cases in 11C-choline PET/CT and in 21 of 78 (27%) cases in BS. Depending on their attribution as either positive or negative, the ranges of sensitivity, specificity, positive predictive value (PPV), negative predictive value (NPV) and accuracy for 11C-choline PET/CT were 89-89%, 98-100%, 96-100%, 94-96% and 95-96%, respectively. For BS they were 100-70%, 75-100%, 68--100%, 100-86% and 83-90%, respectively. Concordant findings between 11C-choline PET/CT and BS occurred in 55 of 78 (71%) cases. Fig. 2. Authors conclude that for its high specificity, 11C-choline PET/CT positive findings may accurately predict the presence of bone metastasis (BM).
11C-Choline PET/CT guided therapies
The promising results largely reported in the use of 11C-choline PET/CT suggested a more extensive use of such technique in the clinical setting. 11C-choline PET/CT can be used to guide aggressive and tailored therapies in selected patients, particularly in those showing few metastatic sites detected by 11C-choline PET/CT. Two different approaches are possible: surgery (performing Pelvic Lymph Node Dissection: PLND) or Radiation Therapy (EBRT, IMRT). The first study evaluating 11C-choline PET/CT as a guide for PLND has been published by Scattoni et al. [17] in 25 prostate cancer patients, who resulted to be positive at 11C-choline PET/CT (21 pts) or at MR (4 pts). Mean PSA value was 1.98 ng/mL. Validation of results was performed by histology after surgery with a mean number of 21.9 nodes per patient removed (range: 4-74). Authors found that 19/21 patients (90%) with positive 11C-choline PET/CT had nodal involvement at histological evaluation, while none of the 4 patients with negative 11C-choline PET/CT had nodal metastases. 11C-choline PET/CT sensitivity, specificity, PPV, NPV and accuracy were 64%, 90%, 86%, 72%, and 77%, respectively. The low NPV seems to depend on the limited capability of 11C-choline PET/CT to detect micro metastases. Very recently Rigatti et al [18] tried to determine whether the removal of positive lymph nodes at 11C-choline PET/CT may have an impact on the prognosis of patients with nodal recurrence after RP. Authors enrolled prospectively 72 patients with no more than 2 positive lymph-nodes at 11C-choline PET/CT. Results are very encouraging since, after a mean follow-up of 39.4 months, the overall biochemical response rate was 56.9%. Preoperative PSA <4 ng/mL and negative lymph nodes at previous RP represented independent predictor of good biochemical response rate. These data confirm, once more, that the early detection of recurrence, when PSA values are low and when metastatic diffusion is supposed to be limited, is crucial to achieve response to treatment. Alongi et al [19] in 2010, for the first time reported a case of 11C-choline PET/CT-guided helical tomotherapy (HTT) concomitant with estramustine therapy for the treatment of nodal recurrence from prostate cancer in one patient. A dose of 67.2Gy was administered in 28 fractions. At 24 months after the end of helical tomotherapy, PSA was undetectable and no late toxicities were recorded. Authors underlined that a disease-free survival of 24 months, in the absence of any type of systemic therapy, is uncommon in metastatic prostate cancer. Very recently Souvatsoglu, et al [20] studied the influence of 11C-choline PET/CT on the extent of the planning target volume (PTV) in thirty-seven patients treated with radical prostatectomy and referred to salvage radiation therapy (RT) to the prostatic fossa because of biochemical relapse. All patients underwent 11C-choline PET/CT before RT. They found that 5/37 patients (13%) had a positive finding in the 11C-choline PET/CT, outside of the prostatic fossa (iliac lymph nodes), implicating an extension of the PTV. Overall, at the 51 months follow-up, 56% of the patients had a PSA < 0.2 ng/mL.
76 y.o. patient. Biochemical relapse 9 years after radical prostatectomy. PSA 1,43 ng/mL at the time of the Bone Scan and 11C-Choline PET/CT. a) The bone scan showed focal uptake in the left clavicle and the right pelvis. b) PET/CT also showed 11C-choline uptake in the same > sites, which were evident on CT (pictures c and d).
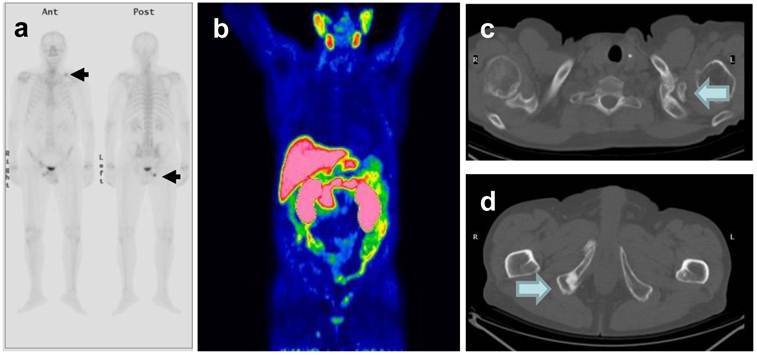
CONCLUSIONS
In summary, considering the previous literature and our own experience, we can conclude that 11C-choline PET/CT could represent an important imaging modality in the detection of distant relapses in prostate cancer patients with biochemical recurrence. According to these data, 11C-choline PET/CT could play a crucial role as first diagnostic procedure in patients at high risk of relapse who demonstrate a fast PSA kinetics, even in presence of low PSA values. 11C-choline PET/CT should be more extensively used mostly in order to address guided therapies such as RT (either EBRT, IMRT or Hormonal Treatment) or surgery with PLND. The results of the few studies published so far are very encouraging, since about half of the patients are free from relapse (either biochemical or clinical) for a significant number of months. This could reduce the use of anti androgenic treatments with a significant benefit to the patients in terms of reduction of side effects and, probably, with a significant reduction in costs. Larger studies are needed to confirm these hypothesis.
Competing Interests
The authors have declared that no competing interest exists.
References
1. ESMO Guidelines Task Force. ESMO minimum clinical recommendations for diagnosis, treatment and follow-up of prostate cancer. Ann Oncol. 2005;16(suppl 1):i34-i36
2. Hricak H, Choyke PL, Eberhardt SC, Leibel SA, Scardino PT. Imaging prostate cancer: a multidisciplinary perspective. Radiology. 2007;243:28-53
3. Cher ML, Bianco FJ Jr, Lam JS, Davis LP, Grignon DJ, Sakr WA, Banerjee M, Pontes JE, Wood DP Jr. Limited role of radionuclide bone scintigraphy in patients with prostate specific antigen elevations after radical prostatectomy. J Urol. 1998;160:1387-91
4. Gomez P, Manoharan M, Kim SS, Soloway MS. Radionuclide bone scintigraphy in patients with biochemical recurrence after radical prostatectomy: when is it indicated? BJU Int. 2004;94:299-302
5. Choueiri TK, Dreicer R, Paciorek A, Carroll PR, Konety B. A model that predicts the probability of positive imaging in prostate cancer cases with biochemical failure after initial definitive local therapy. J Urol. 2008Mar;179(3):906-10
6. Okotie OT, Aronson WJ, Wieder JA, Liao Y, Dorey F, DeKernion JB, Freedland SJ. Predictors of metastatic disease in men with biochemical failure following radical prostatectomy. J Urol. 2004Jun;171(6 Pt 1):2260
7. Phelps ME. Inaugural article: positron emission tomography provides molecular imaging of biological processes. Proc Natl Acad Sci U S A. 2000;97:9226-33
8. Hara T, Kosaka N, Kishi H. PET imaging of prostate cancer using carbon-11-choline. J Nucl Med. 1998;39:990-995
9. Picchio M, Messa C, Landoni C, Gianolli L, Sironi S, Brioschi M, Matarrese M, Matei DV, De Cobelli F, Del Maschio A, Rocco F, Rigatti P, Fazio F. Value of [11C]choline-positron emission tomography for re-staging prostate cancer: a comparison with [18F]fluorodeoxyglucose-positron emission tomography. J Urol. 2003;169:1337-40
10. Krause BJ, Souvatzoglou M, Tuncel M, Herrmann K, Buck AK, Praus C, Schuster T, Geinitz H, Treiber U, Schwaiger M. The detection rate of [(11)C]Choline-PET/CT depends on the serum PSA-value in patients with biochemical recurrence of prostate cancer. Eur J Nucl Med Mol Imaging. 2008;35:18-23
11. Giovacchini G, Picchio M, Coradeschi E, Bettinardi V, Gianolli L, Scattoni V, Cozzarini C, Di Muzio N, Rigatti P, Fazio F, Messa C. Predictive factors of [11C]choline PET/CT in patients with biochemical failure after radical prostatectomy. Eur J Nucl Med Mol Imaging. 2010Feb;37(2):301-9
12. Castellucci P, Fuccio C, Nanni C, Santi I, Rizzello A, Lodi F, Franceschelli A, Martorana G, Manferrati F, Fanti S. Influence of trigger PSA and PSA kinetics on 11C-choline PET/CT detection rate in patients with biochemical relapse after radical prostatectomy. J Nucl Med. 2009Sep;50(9):1394-400
13. Giovacchini G, Picchio M, Scattoni V, Garcia Parra R, Briganti A, Gianolli L, Montorsi F, Messa C. PSA doubling time for prediction of 11C choline PET/CT findings in prostate cancer patients with biochemical failure after radical prostatectomy. Eur J Nucl Med Mol Imaging. 2010Jun;37(6):1106-16
14. Castellucci P, Fuccio C, Rubello D, Schiavina R, Santi I, Nanni C, Allegri V, Montini GC, Ambrosini V, Boschi S, Martorana G, Marzola MC, Fanti S. Is there a role for ¹¹C-choline PET/CT in the early detection of metastatic disease in surgically treated prostate cancer patients with a mild PSA increase <1.5 ng/ml? Eur J Nucl Med Mol Imaging. 2011Jan;38(1):55-63
15. Fuccio C, Castellucci P, Schiavina R, Santi I, Allegri V, Pettinato V, Boschi S, Martorana G, Al-Nahhas A, Rubello D, Fanti S. Role of 11C-choline PET/CT in the restaging of prostate cancer patients showing a single lesion on bone scintigraphy. Ann Nucl Med. 2010;24(6):485-92
16. Picchio M, Spinapolice EG, Fallanca F, Crivellaro C, Giovacchini G, Gianolli L, Messa C. [(11)C]Choline PET/CT detection of bone metastases in patients with PSA progression after primary treatment for prostate cancer: comparison with bone scintigraphy. Eur J Nucl Med Mol Imaging. 2011 epub
17. Scattoni V, Picchio M, Suardi N, Messa C, Freschi M, Roscigno M, Da Pozzo L, Bocciardi A, Rigatti P, Fazio F. Detection of lymph-node metastases with integrated [11C]choline PET/CT in patients with PSA failure after radical retropubic prostatectomy: results confirmed by open pelvic-retroperitoneal lymphadenectomy. Eur Urol. 2007;52:423-9
18. Rigatti P, Suardi N, Briganti A, Da Pozzo LF, Tutolo M, Villa L, Gallina A, Capitanio U, Abdollah F, Scattoni V, Colombo R, Freschi M, Picchio M, Messa C, Guazzoni G, Montorsi F. Pelvic/Retroperitoneal Salvage Lymph Node Dissection for Patients Treated With Radical Prostatectomy With Biochemical Recurrence and Nodal Recurrence Detected by [11C]Choline Positron Emission Tomography/Computed Tomography. Eur Urol. 2011Nov;60(5):935-43
19. Alongi F, Schipani S, Gajate AM, Rosso A, Cozzarini C, Fiorino C, Alongi P, Picchio M, Gianolli L, Messa C, Di Muzio N [11C]choline-PET-guided helical tomotherapy, estramustine in a patient with pelvic-recurrent prostate cancer. local control and toxicity profile after 24 months. Tumori. 2010;96(4):613-7
20. Souvatzoglou M, Krause BJ, Pürschel A, Thamm R, Schuster T, Buck AK, Zimmermann F, Molls M, Schwaiger M, Geinitz H. Influence of (11)C-choline PET/CT on the treatment planning for salvage radiation therapy in patients with biochemical recurrence of prostate cancer. Radiother Oncol. 2011May;99(2):193-200
Author contact
![]() Corresponding author: Paolo Castellucci, MD. & Maria Picchio MD., UO di Medicina Nucleare, PAD. 30, Azienda Ospedaliero-Universitaria di Bologna Policlinico S.Orsola-Malpighi. Via Massarenti, 940138 Bologna , ITALY. Email: paolo.castelluccibo.it Tel.+390516363957 Fax. +390516363956.
Corresponding author: Paolo Castellucci, MD. & Maria Picchio MD., UO di Medicina Nucleare, PAD. 30, Azienda Ospedaliero-Universitaria di Bologna Policlinico S.Orsola-Malpighi. Via Massarenti, 940138 Bologna , ITALY. Email: paolo.castelluccibo.it Tel.+390516363957 Fax. +390516363956.
 Global reach, higher impact
Global reach, higher impact